Ascending midbrain dopaminergic axons require descending GAD65 axon fascicles for normal pathfinding
- PMID: 24926237
- PMCID: PMC4046268
- DOI: 10.3389/fnana.2014.00043
Ascending midbrain dopaminergic axons require descending GAD65 axon fascicles for normal pathfinding
Abstract
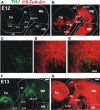
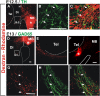
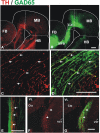
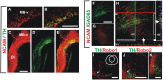
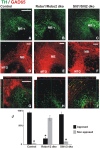
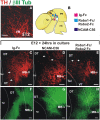
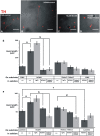
The Nigrostriatal pathway (NSP) is formed by dopaminergic axons that project from the ventral midbrain to the dorsolateral striatum as part of the medial forebrain bundle. Previous studies have implicated chemotropic proteins in the formation of the NSP during development but little is known of the role of substrate-anchored signals in this process. We observed in mouse and rat embryos that midbrain dopaminergic axons ascend in close apposition to descending GAD65-positive axon bundles throughout their trajectory to the striatum. To test whether such interaction is important for dopaminergic axon pathfinding, we analyzed transgenic mouse embryos in which the GAD65 axon bundle was reduced by the conditional expression of the diphtheria toxin. In these embryos we observed dopaminergic misprojection into the hypothalamic region and abnormal projection in the striatum. In addition, analysis of Robo1/2 and Slit1/2 knockout embryos revealed that the previously described dopaminergic misprojection in these embryos is accompanied by severe alterations in the GAD65 axon scaffold. Additional studies with cultured dopaminergic neurons and whole embryos suggest that NCAM and Robo proteins are involved in the interaction of GAD65 and dopaminergic axons. These results indicate that the fasciculation between descending GAD65 axon bundles and ascending dopaminergic axons is required for the stereotypical NSP formation during brain development and that known guidance cues may determine this projection indirectly by instructing the pathfinding of the axons that are part of the GAD65 axon scaffold.
Keywords: NCAM; Robo; axon interaction; dopaminergic; fasciculation; nigrostriatal pathway.
Figures

References
-
- Albanese A., Altavista M. C., Rossi P. (1986). Organization of central nervous system dopaminergic pathways. J. Neural Transm. Suppl. 22, 3–17 - PubMed
-
- Auclair F., Marchand R., Glover J. C. (1999). Regional patterning of reticulospinal and vestibulospinal neurons in the hindbrain of mouse and rat embryos. J. Comp. Neurol. 411, 288–300 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

