Diversity of mitochondria-dependent dilator mechanisms in vascular smooth muscle of cerebral arteries from normal and insulin-resistant rats
- PMID: 24929852
- PMCID: PMC4137120
- DOI: 10.1152/ajpheart.00091.2014
Diversity of mitochondria-dependent dilator mechanisms in vascular smooth muscle of cerebral arteries from normal and insulin-resistant rats
Abstract
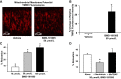
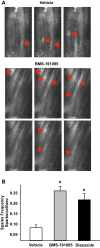
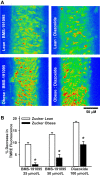
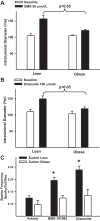
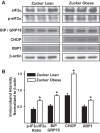
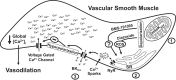
Mitochondrial depolarization following ATP-sensitive potassium (mitoKATP) channel activation has been shown to induce cerebral vasodilation by generation of mitochondrial reactive oxygen species (ROS), which sequentially promotes frequency of calcium sparks and activation of large conductance calcium-activated potassium channels (BKCa) in vascular smooth muscle (VSM). We previously demonstrated that cerebrovascular insulin resistance accompanies aging and obesity. It is unclear whether mitochondrial depolarization without the ROS generation enhances calcium sparks and vasodilation in phenotypically normal [Sprague Dawley (SD); Zucker lean (ZL)] and insulin-resistant [Zucker obese (ZO)] rats. We compared the mechanisms underlying the vasodilation to ROS-dependent (diazoxide) and ROS-independent [BMS-191095 (BMS)] mitoKATP channel activators in normal and ZO rats. Arterial diameter studies from SD, ZL, and ZO rats showed that BMS as well as diazoxide induced vasodilation in endothelium-denuded cerebral arteries. In normal rats, BMS-induced vasodilation was mediated by mitochondrial depolarization and calcium sparks generation in VSM and was reduced by inhibition of BKCa channels. However, unlike diazoxide-induced vasodilation, scavenging of ROS had no effect on BMS-induced vasodilation. Electron spin resonance spectroscopy confirmed that diazoxide but not BMS promoted vascular ROS generation. BMS- as well as diazoxide-induced vasodilation, mitochondrial depolarization, and calcium spark generation were diminished in cerebral arteries from ZO rats. Thus pharmacological depolarization of VSM mitochondria by BMS promotes ROS-independent vasodilation via generation of calcium sparks and activation of BKCa channels. Diminished generation of calcium sparks and reduced vasodilation in ZO arteries in response to BMS and diazoxide provide new insights into mechanisms of cerebrovascular dysfunction in insulin resistance.
Figures

References
-
- Ahmad N, Wang Y, Haider KH, Wang B, Pasha Z, Uzun O, Ashraf M. Cardiac protection by mitoKATP channels is dependent on Akt translocation from cytosol to mitochondria during late preconditioning. Am J Physiol Heart Circ Physiol 290: H2402–H2408, 2006 - PubMed
-
- Bray GA. The Zucker-fatty rat: a review. Fed Proc 36: 148–153, 1977 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

