Insulin-like growth factor 2 silencing restores taxol sensitivity in drug resistant ovarian cancer
- PMID: 24932685
- PMCID: PMC4059749
- DOI: 10.1371/journal.pone.0100165
Insulin-like growth factor 2 silencing restores taxol sensitivity in drug resistant ovarian cancer
Abstract
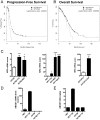
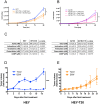
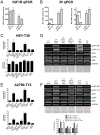
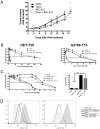
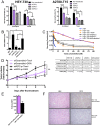
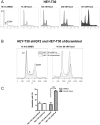
Drug resistance is an obstacle to the effective treatment of ovarian cancer. We and others have shown that the insulin-like growth factor (IGF) signaling pathway is a novel potential target to overcome drug resistance. The purpose of this study was to validate IGF2 as a potential therapeutic target in drug resistant ovarian cancer and to determine the efficacy of targeting IGF2 in vivo. An analysis of The Cancer Genome Atlas (TCGA) data in the serous ovarian cancer cohort showed that high IGF2 mRNA expression is significantly associated with shortened interval to disease progression and death, clinical indicators of drug resistance. In a genetically diverse panel of ovarian cancer cell lines, the IGF2 mRNA levels measured in cell lines resistant to various microtubule-stabilizing agents including Taxol were found to be significantly elevated compared to the drug sensitive cell lines. The effect of IGF2 knockdown on Taxol resistance was investigated in vitro and in vivo. Transient IGF2 knockdown significantly sensitized drug resistant cells to Taxol treatment. A Taxol-resistant ovarian cancer xenograft model, developed from HEY-T30 cells, exhibited extreme drug resistance, wherein the maximal tolerated dose of Taxol did not delay tumor growth in mice. Blocking the IGF1R (a transmembrane receptor that transmits signals from IGF1 and IGF2) using a monoclonal antibody did not alter the response to Taxol. However, stable IGF2 knockdown using short-hairpin RNA in HEY-T30 effectively restored Taxol sensitivity. These findings validate IGF2 as a potential therapeutic target in drug resistant ovarian cancer and show that directly targeting IGF2 may be a preferable strategy compared with targeting IGF1R alone.
Conflict of interest statement
Figures
Similar articles
-
Insulin-like growth factor 2 expression modulates Taxol resistance and is a candidate biomarker for reduced disease-free survival in ovarian cancer.Clin Cancer Res. 2010 Jun 1;16(11):2999-3010. doi: 10.1158/1078-0432.CCR-09-3233. Epub 2010 Apr 19. Clin Cancer Res. 2010. PMID: 20404007 Free PMC article.
-
Upregulation of phosphorylated cofilin 1 correlates with taxol resistance in human ovarian cancer in vitro and in vivo.Oncol Rep. 2013 Jan;29(1):58-66. doi: 10.3892/or.2012.2078. Epub 2012 Oct 12. Oncol Rep. 2013. PMID: 23064469
-
Impact of constitutive IGF1/IGF2 stimulation on the transcriptional program of human breast cancer cells.Carcinogenesis. 2007 Jan;28(1):49-59. doi: 10.1093/carcin/bgl091. Epub 2006 Jun 14. Carcinogenesis. 2007. PMID: 16774935
-
Targeting the insulin growth factor receptor 1.Hematol Oncol Clin North Am. 2012 Jun;26(3):527-42, vii-viii. doi: 10.1016/j.hoc.2012.01.004. Epub 2012 Feb 28. Hematol Oncol Clin North Am. 2012. PMID: 22520978 Free PMC article. Review.
-
Autophagy in cancer resistance to paclitaxel: Development of combination strategies.Biomed Pharmacother. 2023 May;161:114458. doi: 10.1016/j.biopha.2023.114458. Epub 2023 Mar 6. Biomed Pharmacother. 2023. PMID: 36889112 Review.
Cited by
-
The Interplay Between Non-coding RNAs and Insulin-Like Growth Factor Signaling in the Pathogenesis of Neoplasia.Front Cell Dev Biol. 2021 Mar 9;9:634512. doi: 10.3389/fcell.2021.634512. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33768092 Free PMC article. Review.
-
Exosomal miR-543 Inhibits the Proliferation of Ovarian Cancer by Targeting IGF2.J Immunol Res. 2022 Mar 29;2022:2003739. doi: 10.1155/2022/2003739. eCollection 2022. J Immunol Res. 2022. PMID: 35391781 Free PMC article.
-
Insulin-like growth factor 2 axis supports the serum-independent growth of malignant rhabdoid tumor and is activated by microenvironment stress.Oncotarget. 2017 Jul 18;8(29):47269-47283. doi: 10.18632/oncotarget.17617. Oncotarget. 2017. PMID: 28521298 Free PMC article.
-
Insulin-like growth factor (IGF) signaling in tumorigenesis and the development of cancer drug resistance.Genes Dis. 2015 Mar 1;2(1):13-25. doi: 10.1016/j.gendis.2014.10.004. Genes Dis. 2015. PMID: 25984556 Free PMC article.
-
IGF2 Mediates Resistance to Isoform-Selective-Inhibitors of the PI3K in HPV Positive Head and Neck Cancer.Cancers (Basel). 2021 May 7;13(9):2250. doi: 10.3390/cancers13092250. Cancers (Basel). 2021. PMID: 34067117 Free PMC article.
References
-
- Baker J, Liu JP, Robertson EJ, Efstratiadis A (1993) Role of insulin-like growth factors in embryonic and postnatal growth. Cell 75: 73–82. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

