A translocated effector required for Bartonella dissemination from derma to blood safeguards migratory host cells from damage by co-translocated effectors
- PMID: 24945914
- PMCID: PMC4063953
- DOI: 10.1371/journal.ppat.1004187
A translocated effector required for Bartonella dissemination from derma to blood safeguards migratory host cells from damage by co-translocated effectors
Abstract
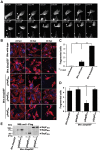
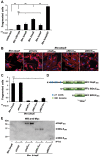
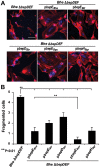
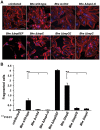
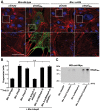
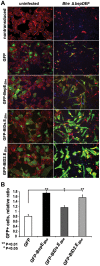
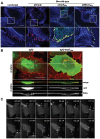
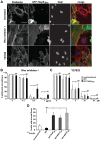
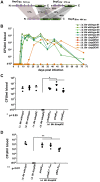
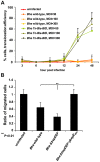
Numerous bacterial pathogens secrete multiple effectors to modulate host cellular functions. These effectors may interfere with each other to efficiently control the infection process. Bartonellae are Gram-negative, facultative intracellular bacteria using a VirB type IV secretion system to translocate a cocktail of Bartonella effector proteins (Beps) into host cells. Based on in vitro infection models we demonstrate here that BepE protects infected migratory cells from injurious effects triggered by BepC and is required for in vivo dissemination of bacteria from the dermal site of inoculation to blood. Human endothelial cells (HUVECs) infected with a ΔbepE mutant of B. henselae (Bhe) displayed a cell fragmentation phenotype resulting from Bep-dependent disturbance of rear edge detachment during migration. A ΔbepCE mutant did not show cell fragmentation, indicating that BepC is critical for triggering this deleterious phenotype. Complementation of ΔbepE with BepEBhe or its homologues from other Bartonella species abolished cell fragmentation. This cyto-protective activity is confined to the C-terminal Bartonella intracellular delivery (BID) domain of BepEBhe (BID2.EBhe). Ectopic expression of BID2.EBhe impeded the disruption of actin stress fibers by Rho Inhibitor 1, indicating that BepE restores normal cell migration via the RhoA signaling pathway, a major regulator of rear edge retraction. An intradermal (i.d.) model for B. tribocorum (Btr) infection in the rat reservoir host mimicking the natural route of infection by blood sucking arthropods allowed demonstrating a vital role for BepE in bacterial dissemination from derma to blood. While the Btr mutant ΔbepDE was abacteremic following i.d. inoculation, complementation with BepEBtr, BepEBhe or BIDs.EBhe restored bacteremia. Given that we observed a similar protective effect of BepEBhe on infected bone marrow-derived dendritic cells migrating through a monolayer of lymphatic endothelial cells we propose that infected dermal dendritic cells may be involved in disseminating Bartonella towards the blood stream in a BepE-dependent manner.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Comment in
-
Bacterial pathogenesis: Bartonella gets under the skin.Nat Rev Microbiol. 2014 Aug;12(8):529. doi: 10.1038/nrmicro3321. Nat Rev Microbiol. 2014. PMID: 25029021 No abstract available.
Similar articles
-
Bartonella type IV secretion effector BepC induces stress fiber formation through activation of GEF-H1.PLoS Pathog. 2021 Jan 28;17(1):e1009065. doi: 10.1371/journal.ppat.1009065. eCollection 2021 Jan. PLoS Pathog. 2021. PMID: 33508039 Free PMC article.
-
Bartonella effector protein C mediates actin stress fiber formation via recruitment of GEF-H1 to the plasma membrane.PLoS Pathog. 2021 Jan 28;17(1):e1008548. doi: 10.1371/journal.ppat.1008548. eCollection 2021 Jan. PLoS Pathog. 2021. PMID: 33508040 Free PMC article.
-
Combined action of the type IV secretion effector proteins BepC and BepF promotes invasome formation of Bartonella henselae on endothelial and epithelial cells.Cell Microbiol. 2011 Feb;13(2):284-99. doi: 10.1111/j.1462-5822.2010.01535.x. Epub 2010 Oct 22. Cell Microbiol. 2011. PMID: 20964799
-
New insights into the role of Bartonella effector proteins in pathogenesis.Curr Opin Microbiol. 2015 Feb;23:80-5. doi: 10.1016/j.mib.2014.11.007. Epub 2014 Nov 26. Curr Opin Microbiol. 2015. PMID: 25461577 Review.
-
Role of distinct type-IV-secretion systems and secreted effector sets in host adaptation by pathogenic Bartonella species.Cell Microbiol. 2019 Mar;21(3):e13004. doi: 10.1111/cmi.13004. Epub 2019 Feb 6. Cell Microbiol. 2019. PMID: 30644157 Free PMC article. Review.
Cited by
-
Bartonella type IV secretion effector BepC induces stress fiber formation through activation of GEF-H1.PLoS Pathog. 2021 Jan 28;17(1):e1009065. doi: 10.1371/journal.ppat.1009065. eCollection 2021 Jan. PLoS Pathog. 2021. PMID: 33508039 Free PMC article.
-
Cutting Edge: Redundant Roles for MHC Class II-, CD1d-, and MR1-restricted T Cells in Clearing Bartonella Infection.J Immunol. 2024 Sep 1;213(5):553-558. doi: 10.4049/jimmunol.2400045. J Immunol. 2024. PMID: 38984869 Free PMC article.
-
Versatility of the BID Domain: Conserved Function as Type-IV-Secretion-Signal and Secondarily Evolved Effector Functions Within Bartonella-Infected Host Cells.Front Microbiol. 2019 May 3;10:921. doi: 10.3389/fmicb.2019.00921. eCollection 2019. Front Microbiol. 2019. PMID: 31130928 Free PMC article. Review.
-
The Impact of Bartonella VirB/VirD4 Type IV Secretion System Effectors on Eukaryotic Host Cells.Front Microbiol. 2021 Dec 15;12:762582. doi: 10.3389/fmicb.2021.762582. eCollection 2021. Front Microbiol. 2021. PMID: 34975788 Free PMC article. Review.
-
The BID Domain of Type IV Secretion Substrates Forms a Conserved Four-Helix Bundle Topped with a Hook.Structure. 2017 Jan 3;25(1):203-211. doi: 10.1016/j.str.2016.10.010. Epub 2016 Nov 23. Structure. 2017. PMID: 27889208 Free PMC article.
References
-
- Monack DM, Mueller A, Falkow S (2004) Persistent bacterial infections: the interface of the pathogen and the host immune system. Nat Rev Microbiol 2: 747–765. - PubMed
-
- Kido M, Watanabe N, Aoki N, Iwamoto S, Nishiura H, et al. (2011) Dual roles of CagA protein in Helicobacterpylori-induced chronic gastritis in mice. Biochem Biophys Res Commun 412: 266–272. - PubMed
-
- Diacovich L, Gorvel JP (2010) Bacterial manipulation of innate immunity to promote infection. Nat Rev Microbiol 8: 117–128. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

