Assessment of packed bed bioreactor systems in the production of viral vaccines
- PMID: 24949260
- PMCID: PMC4052670
- DOI: 10.1186/s13568-014-0025-z
Assessment of packed bed bioreactor systems in the production of viral vaccines
Abstract
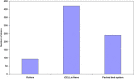
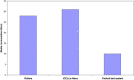
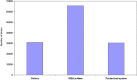
Vaccination is believed to be the most effective method for the prevention of infectious diseases. Thus it is imperative to develop cost effective and scalable process for the production of vaccines so as to make them affordable for mass use. In this study, performance of a novel disposable iCELLis fixed bed bioreactor system was investigated for the production of some viral vaccines like Rabies, Hepatitis-A and Chikungunya vaccines in comparison to conventional systems like the commercially available packed bed system and roller bottle system. Vero and MRC-5 cell substrates were evaluated for growth parameters in all the three systems maintaining similar seeding density, multiplicity of infection (MOI) and media components. It was observed that Vero cells showed similar growth in all the three bioreactors whereas MRC-5 cells showed better growth in iCELLis Nano system and roller bottle system. Subsequently, the virus infection and antigen production studies also revealed that for Hepatitis-A and Chikungunya iCELLis Nano bioreactor system was better to the commercial packed bed bioreactor and roller bottle systems. Although for rabies antigen production commercially available packed bed bioreactor system was found to be better. This study shows that different bioreactor platforms may be employed for viral vaccine production and iCELLis Nano is one of such new convenient and a stable platform for production of human viral vaccines.
Keywords: Chikungunya virus; Hepatitis-A virus; Packed bed bioreactor; Rabies virus; Vaccine production; iCELLis Nano bioreactor.
Figures

References
-
- Aunins JG. In: Encylopedia of cell technology. Spier ER, editor. Wiley, New York; 2000. Viral vaccine production in cell culture; pp. 1183–1207.
-
- Butler M. Growth limitations on microcarriers. Adv Biochem Eng Biotechnol. 1987;4:57–84. - PubMed
-
- Drugmand JC, Havelange N, Debras F, Collignon F, Mathieu E, Castillo J. Vaccine production in iCELLis fixed bed disposable bioreactor. Veterinary Vaccines Düsseldorf, Germany; 2009.
-
- Drugmand JC, Dubois S, Dohogne Y, Havelange N, Castillo J. Viral vaccine production at manufacturing size into iCELLisTM disposable fixed-bed reactor. Vaccine Production Summit, La Jolla, CA, USA; 2010.
LinkOut - more resources
Full Text Sources
Other Literature Sources

