Integrative gene transfer in the truffle Tuber borchii by Agrobacterium tumefaciens-mediated transformation
- PMID: 24949275
- PMCID: PMC4052689
- DOI: 10.1186/s13568-014-0043-x
Integrative gene transfer in the truffle Tuber borchii by Agrobacterium tumefaciens-mediated transformation
Abstract
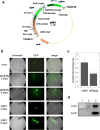
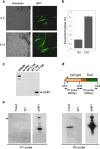
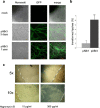
Agrobacterium tumefaciens-mediated transformation is a powerful tool for reverse genetics and functional genomic analysis in a wide variety of plants and fungi. Tuber spp. are ecologically important and gastronomically prized fungi ("truffles") with a cryptic life cycle, a subterranean habitat and a symbiotic, but also facultative saprophytic lifestyle. The genome of a representative member of this group of fungi has recently been sequenced. However, because of their poor genetic tractability, including transformation, truffles have so far eluded in-depth functional genomic investigations. Here we report that A. tumefaciens can infect Tuber borchii mycelia, thereby conveying its transfer DNA with the production of stably integrated transformants. We constructed two new binary plasmids (pABr1 and pABr3) and tested them as improved transformation vectors using the green fluorescent protein as reporter gene and hygromycin phosphotransferase as selection marker. Transformants were stable for at least 12 months of in vitro culture propagation and, as revealed by TAIL- PCR analysis, integration sites appear to be heterogeneous, with a preference for repeat element-containing genome sites.
Keywords: Agrobacterium tumefaciens-mediated transformation; Green fluorescent protein; Hygromycin phosphotransferase; T-DNA; TAIL-PCR; Truffles; Tuber spp; binary plasmid.
Figures

Similar articles
-
Agrobacterium-mediated gene transfer and enhanced green fluorescent protein visualization in the mycorrhizal ascomycete Tuber borchii: a first step towards truffle genetics.Curr Genet. 2005 Jul;48(1):69-74. doi: 10.1007/s00294-005-0579-z. Epub 2005 May 3. Curr Genet. 2005. PMID: 15868150
-
Agrobacterium- tumefaciens-mediated transformation of Helminthosporium turcicum, the maize leaf-blight fungus.Arch Microbiol. 2003 Oct;180(4):279-84. doi: 10.1007/s00203-003-0589-5. Epub 2003 Jul 31. Arch Microbiol. 2003. PMID: 12898134
-
Agrobacterium-Mediated Transformation of Fusarium oxysporum: An Efficient Tool for Insertional Mutagenesis and Gene Transfer.Phytopathology. 2001 Feb;91(2):173-80. doi: 10.1094/PHYTO.2001.91.2.173. Phytopathology. 2001. PMID: 18944391
-
Current state and perspectives of truffle genetics and sustainable biotechnology.Appl Microbiol Biotechnol. 2006 Sep;72(3):437-41. doi: 10.1007/s00253-006-0519-y. Epub 2006 Jun 27. Appl Microbiol Biotechnol. 2006. PMID: 16802150 Review.
-
A guide to binary vectors and strategies for targeted genome modification in fungi using Agrobacterium tumefaciens-mediated transformation.J Microbiol Methods. 2011 Dec;87(3):247-62. doi: 10.1016/j.mimet.2011.09.004. Epub 2011 Sep 17. J Microbiol Methods. 2011. PMID: 21951772 Review.
Cited by
-
Tuber borchii Shapes the Ectomycorrhizosphere Microbial Communities of Corylus avellana.Mycobiology. 2019 Jun 17;47(2):180-190. doi: 10.1080/12298093.2019.1615297. eCollection 2019. Mycobiology. 2019. PMID: 31448138 Free PMC article.
-
Agrobacterium tumefaciens-Mediated Genetic Transformation of the Ect-endomycorrhizal Fungus Terfezia boudieri.Genes (Basel). 2020 Oct 30;11(11):1293. doi: 10.3390/genes11111293. Genes (Basel). 2020. PMID: 33143066 Free PMC article.
References
-
- Ambra R, Grimaldi B, Zamboni S, Filetici P, Macino G, Ballario P. Photomorphogenesis in the hypogeous fungus Tuber borchii: isolation and characterization of Tbwc-1, the homologue of the blue-light photoreceptor of Neurospora crassa. Fungal Genet Biol. 2004;4(7):688–697. doi: 10.1016/j.fgb.2004.02.004. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

