Fourier ptychographic microscopy for filtration-based circulating tumor cell enumeration and analysis
- PMID: 24949708
- PMCID: PMC4572097
- DOI: 10.1117/1.JBO.19.6.066007
Fourier ptychographic microscopy for filtration-based circulating tumor cell enumeration and analysis
Erratum in
-
Errata: Fourier ptychographic microscopy for filtration-based circulating tumor cell enumeration and analysis.J Biomed Opt. 2015 Dec;20(12):129802. doi: 10.1117/1.JBO.20.12.129802. J Biomed Opt. 2015. PMID: 26677072 Free PMC article. No abstract available.
Abstract
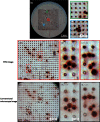
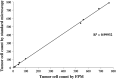
Circulating tumor cells (CTCs) are recognized as a candidate biomarker with strong prognostic and predictive potential in metastatic disease. Filtration-based enrichment technologies have been used for CTC characterization, and our group has previously developed a membrane microfilter device that demonstrates efficacy in model systems and clinical blood samples. However, uneven filtration surfaces make the use of standard microscopic techniques a difficult task, limiting the performance of automated imaging using commercially available technologies. Here, we report the use of Fourier ptychographic microscopy (FPM) to tackle this challenge. Employing this method, we were able to obtain high-resolution color images, including amplitude and phase, of the microfilter samples over large areas. FPM's ability to perform digital refocusing on complex images is particularly useful in this setting as, in contrast to other imaging platforms, we can focus samples on multiple focal planes within the same frame despite surface unevenness. In model systems, FPM demonstrates high image quality, efficiency, and consistency in detection of tumor cells when comparing corresponding microfilter samples to standard microscopy with high correlation (R² = 0.99932). Based on these results, we believe that FPM will have important implications for improved, high throughput, filtration-based CTC analysis, and, more generally, image analysis of uneven surfaces.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

