A polymorphism in JMJD2C alters the cleavage by caspase-3 and the prognosis of human breast cancer
- PMID: 24952432
- PMCID: PMC4148098
- DOI: 10.18632/oncotarget.2029
A polymorphism in JMJD2C alters the cleavage by caspase-3 and the prognosis of human breast cancer
Abstract
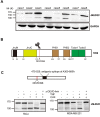
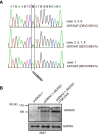
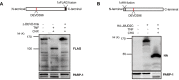
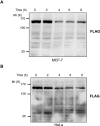
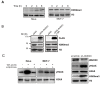
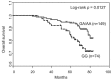
JMJD2C is a candidate oncogene that encodes a histone lysine demethylase with the ability to demethylate the lysine 9 residue of histone H3 (H3K9). The expression levels of JMJD2C are associated with tumor development and clinical outcome. Here we identify JMJD2C as a new substrate for caspase-3. JMJD2C is cleaved by caspase-3 at DEVD396G motif and then loses its demethylase activity. Additionally, we uncover D396N polymorphism (rs2296067) in the cleavage site of JMJD2C and establish its influence on the resistant to the cleavage by caspase-3. Importantly, we determined that D396N polymorphism is significantly associated with the prognosis of human breast cancer. We further found that the basal levels of DSB (double strand DNA break) repair proteins γ-H2AX (gamma-H2AX) increased when cells were treated with tumor necrosis factor-α (TNF-α) which activates caspase-3 activity. We also show that knockdown of JMJD2C expression results in up-regulation of basal γ-H2AX. We propose that D396N polymorphism of JMJD2C affects the prognosis of human breast cancer via altering the cleavage by caspase-3 and the ability of DSB repair which may contribute to therapy resistance.
Figures
Similar articles
-
Histone demethylase JMJD2C is a coactivator for hypoxia-inducible factor 1 that is required for breast cancer progression.Proc Natl Acad Sci U S A. 2012 Dec 4;109(49):E3367-76. doi: 10.1073/pnas.1217394109. Epub 2012 Nov 5. Proc Natl Acad Sci U S A. 2012. PMID: 23129632 Free PMC article.
-
Cooperative demethylation by JMJD2C and LSD1 promotes androgen receptor-dependent gene expression.Nat Cell Biol. 2007 Mar;9(3):347-53. doi: 10.1038/ncb1546. Epub 2007 Feb 4. Nat Cell Biol. 2007. PMID: 17277772
-
Jmjd2c histone demethylase enhances the expression of Mdm2 oncogene.Biochem Biophys Res Commun. 2009 Nov 13;389(2):366-71. doi: 10.1016/j.bbrc.2009.08.155. Epub 2009 Sep 2. Biochem Biophys Res Commun. 2009. PMID: 19732750
-
Histone demethylase JMJD2C: epigenetic regulators in tumors.Oncotarget. 2017 Jul 12;8(53):91723-91733. doi: 10.18632/oncotarget.19176. eCollection 2017 Oct 31. Oncotarget. 2017. PMID: 29207681 Free PMC article. Review.
-
Histone demethylase lysine demethylase 5B in development and cancer.Oncotarget. 2017 Jan 31;8(5):8980-8991. doi: 10.18632/oncotarget.13858. Oncotarget. 2017. PMID: 27974677 Free PMC article. Review.
Cited by
-
The precision relationships between eight GWAS-identified genetic variants and breast cancer in a Chinese population.Oncotarget. 2016 Nov 15;7(46):75457-75467. doi: 10.18632/oncotarget.12255. Oncotarget. 2016. PMID: 27705907 Free PMC article.
-
Associations of Caspase-3 gene polymorphism with lumbar disc herniation.Kaohsiung J Med Sci. 2016 Nov;32(11):552-558. doi: 10.1016/j.kjms.2016.08.010. Epub 2016 Oct 14. Kaohsiung J Med Sci. 2016. PMID: 27847097 Free PMC article.
-
Histone demethylase JMJD2B and JMJD2C induce fibroblast growth factor 2: mediated tumorigenesis of osteosarcoma.Med Oncol. 2015 Mar;32(3):53. doi: 10.1007/s12032-015-0503-4. Epub 2015 Jan 31. Med Oncol. 2015. PMID: 25636512
-
Jmjd2c maintains the ALDHbri+ cancer stemness with transcription factor SOX2 in lung squamous cell carcinoma.Cancer Biol Ther. 2024 Dec 31;25(1):2373447. doi: 10.1080/15384047.2024.2373447. Epub 2024 Jul 8. Cancer Biol Ther. 2024. PMID: 38975736 Free PMC article.
-
TAB3 O-GlcNAcylation promotes metastasis of triple negative breast cancer.Oncotarget. 2016 Apr 19;7(16):22807-18. doi: 10.18632/oncotarget.8182. Oncotarget. 2016. PMID: 27009840 Free PMC article.
References
-
- Cloos PA, Christensen J, Agger K, Maiolica A, Rappsilber J, Antal T, Hansen KH, Helin K. The putative oncogene GASC1 demethylates tri- and dimethylated lysine 9 on histone H3. Nature. 2006;442(7100):307–311. - PubMed
-
- Yang ZQ, Imoto I, Fukuda Y, Pimkhaokham A, Shimada Y, Imamura M, Sugano S, Nakamura Y, Inazawa J. Identification of a novel gene, GASC1, within an amplicon at 9p23-24 frequently detected in esophageal cancer cell lines. Cancer research. 2000;60(17):4735–4739. - PubMed
-
- Zhao Y, Thomas HD, Batey MA, Cowell IG, Richardson CJ, Griffin RJ, Calvert AH, Newell DR, Smith GC, Curtin NJ. Preclinical evaluation of a potent novel DNA-dependent protein kinase inhibitor NU7441. Cancer research. 2006;66(10):5354–5362. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

