Functional consequences of cocaine re-exposure after discontinuation of cocaine availability
- PMID: 24953829
- PMCID: PMC4132869
- DOI: 10.1016/j.neuropharm.2014.06.003
Functional consequences of cocaine re-exposure after discontinuation of cocaine availability
Abstract
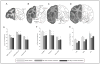
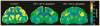
Cocaine users exhibit a wide range of behavioral impairments accompanied by brain structural, neurochemical and functional abnormalities. Metabolic mapping studies in cocaine users and animal models have shown extensive functional alterations throughout the striatum, limbic system, and cortex. Few studies, however, have evaluated the persistence of these effects following cessation of cocaine availability. The purpose of this study, therefore, was to assess the functional effects of re-exposure to cocaine in nonhuman primates after the discontinuation of cocaine self-administration for 30 or 90 days, using the quantitative autoradiographic 2-[14C]deoxyglucose (2DG) method. Rhesus monkeys self-administered cocaine (fixed interval 3-min schedule, 30 infusions per session, 0.3 mg/kg/infusion) for 100 sessions followed by 30 (n=4) or 90 days (n=3) during which experimental sessions were not conducted. Food-reinforced control animals (n=5) underwent identical schedules of reinforcement. Animals were then re-exposed to cocaine or food for one final session and the 2DG method applied immediately after session completion. Compared to controls, re-exposure to cocaine after 30 or 90 day drug-free periods resulted in lower rates of glucose utilization in ventral and dorsal striatum, prefrontal and temporal cortex, limbic system, thalamus, and midbrain. These data demonstrate that vulnerability to the effects of cocaine persists for as long as 90 days after cessation of drug use. While there was some evidence for recovery (fewer brain areas were affected by cocaine re-exposure at 90 days as compared to 30 days), this was not uniform across regions, thus suggesting that recovery occurs at different rates in different brain systems.
Keywords: Cocaine; Functional brain activity; Re-exposure; Rhesus monkey; Self-administration.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures
References
-
- Amaral DG, Price JL, Pitkanen A, Carmichael ST. Anatomical organization of the primate amygdaloid complex. Wiley-Liss; New York: 1992.
-
- Baker DA, McFarland K, Lake RW, Shen H, Tang XC, Toda S, Kalivas PW. Neuroadaptations in cystine-glutamate exchange underlie cocaine relapse. Nat Neurosci. 2003;6:743–749. - PubMed
-
- Baker DA, Shen H, Kalivas PW. Cystine/glutamate exchange serves as the source for extracellular glutamate: modifications by repeated cocaine administration. Amino Acids. 2002;23:161–162. - PubMed
-
- Bechara A, Damasio H, Damasio AR. Emotion, decision making and the orbitofrontal cortex. Cereb Cortex. 2000;10:295–307. - PubMed
-
- Berry J, van Gorp WG, Herzberg DS, Hinkin C, Boone K, Steinman L, Wilkins JN. Neuropsychological deficits in abstinent cocaine abusers: preliminary findings after two weeks of abstinence. Drug Alcohol Depend. 1993;32:231–237. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

