Yeast Dun1 kinase regulates ribonucleotide reductase inhibitor Sml1 in response to iron deficiency
- PMID: 24958100
- PMCID: PMC4135553
- DOI: 10.1128/MCB.00472-14
Yeast Dun1 kinase regulates ribonucleotide reductase inhibitor Sml1 in response to iron deficiency
Abstract
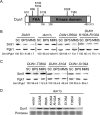
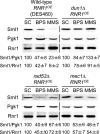
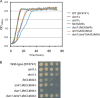
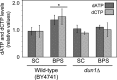
Iron is an essential micronutrient for all eukaryotic organisms because it participates as a redox-active cofactor in many biological processes, including DNA replication and repair. Eukaryotic ribonucleotide reductases (RNRs) are Fe-dependent enzymes that catalyze deoxyribonucleoside diphosphate (dNDP) synthesis. We show here that the levels of the Sml1 protein, a yeast RNR large-subunit inhibitor, specifically decrease in response to both nutritional and genetic Fe deficiencies in a Dun1-dependent but Mec1/Rad53- and Aft1-independent manner. The decline of Sml1 protein levels upon Fe starvation depends on Dun1 forkhead-associated and kinase domains, the 26S proteasome, and the vacuolar proteolytic pathway. Depletion of core components of the mitochondrial iron-sulfur cluster assembly leads to a Dun1-dependent diminution of Sml1 protein levels. The physiological relevance of Sml1 downregulation by Dun1 under low-Fe conditions is highlighted by the synthetic growth defect observed between dun1Δ and fet3Δ fet4Δ mutants, which is rescued by SML1 deletion. Consistent with an increase in RNR function, Rnr1 protein levels are upregulated upon Fe deficiency. Finally, dun1Δ mutants display defects in deoxyribonucleoside triphosphate (dNTP) biosynthesis under low-Fe conditions. Taken together, these results reveal that the Dun1 checkpoint kinase promotes RNR function in response to Fe starvation by stimulating Sml1 protein degradation.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures




Similar articles
-
Yeast Dun1 Kinase Regulates Ribonucleotide Reductase Small Subunit Localization in Response to Iron Deficiency.J Biol Chem. 2016 Apr 29;291(18):9807-17. doi: 10.1074/jbc.M116.720862. Epub 2016 Mar 12. J Biol Chem. 2016. PMID: 26970775 Free PMC article.
-
Ixr1 is required for the expression of the ribonucleotide reductase Rnr1 and maintenance of dNTP pools.PLoS Genet. 2011 May;7(5):e1002061. doi: 10.1371/journal.pgen.1002061. Epub 2011 May 5. PLoS Genet. 2011. PMID: 21573136 Free PMC article.
-
The Dun1 checkpoint kinase phosphorylates and regulates the ribonucleotide reductase inhibitor Sml1.Proc Natl Acad Sci U S A. 2002 Mar 19;99(6):3746-51. doi: 10.1073/pnas.062502299. Proc Natl Acad Sci U S A. 2002. PMID: 11904430 Free PMC article.
-
Rnr1's role in telomere elongation cannot be replaced by Rnr3: a role beyond dNTPs?Curr Genet. 2018 Jun;64(3):547-550. doi: 10.1007/s00294-017-0779-3. Epub 2017 Nov 8. Curr Genet. 2018. PMID: 29119271 Review.
-
Function and regulation of yeast ribonucleotide reductase: cell cycle, genotoxic stress, and iron bioavailability.Biomed J. 2013 Mar-Apr;36(2):51-8. doi: 10.4103/2319-4170.110398. Biomed J. 2013. PMID: 23644233 Review.
Cited by
-
Phosphorylation and Proteasome Recognition of the mRNA-Binding Protein Cth2 Facilitates Yeast Adaptation to Iron Deficiency.mBio. 2018 Sep 18;9(5):e01694-18. doi: 10.1128/mBio.01694-18. mBio. 2018. PMID: 30228242 Free PMC article.
-
Synergistic Effect of a Combination of Proteasome and Ribonucleotide Reductase Inhibitors in a Biochemical Model of the Yeast Saccharomyces cerevisiae and a Glioblastoma Cell Line.Int J Mol Sci. 2024 Apr 3;25(7):3977. doi: 10.3390/ijms25073977. Int J Mol Sci. 2024. PMID: 38612788 Free PMC article.
-
Etp1 confers arsenite resistance by affecting ACR3 expression.FEMS Yeast Res. 2022 Apr 26;22(1):foac018. doi: 10.1093/femsyr/foac018. FEMS Yeast Res. 2022. PMID: 35323907 Free PMC article.
-
Yeast Dun1 Kinase Regulates Ribonucleotide Reductase Small Subunit Localization in Response to Iron Deficiency.J Biol Chem. 2016 Apr 29;291(18):9807-17. doi: 10.1074/jbc.M116.720862. Epub 2016 Mar 12. J Biol Chem. 2016. PMID: 26970775 Free PMC article.
-
Sterol Composition Modulates the Response of Saccharomyces cerevisiae to Iron Deficiency.J Fungi (Basel). 2021 Oct 25;7(11):901. doi: 10.3390/jof7110901. J Fungi (Basel). 2021. PMID: 34829190 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
