Arsenic induces sustained impairment of skeletal muscle and muscle progenitor cell ultrastructure and bioenergetics
- PMID: 24960579
- PMCID: PMC4159748
- DOI: 10.1016/j.freeradbiomed.2014.06.012
Arsenic induces sustained impairment of skeletal muscle and muscle progenitor cell ultrastructure and bioenergetics
Abstract
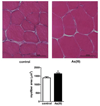
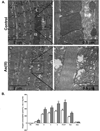
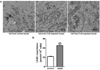
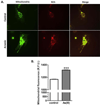
Over 4 million individuals in the United States, and over 140 million individuals worldwide, are exposed daily to arsenic-contaminated drinking water. Human exposures can range from below the current limit of 10 μg/L to over 1mg/L, with 100 μg/L promoting disease in a large portion of those exposed. Although increased attention has recently been paid to myopathy following arsenic exposure, the pathogenic mechanisms underlying clinical symptoms remain poorly understood. This study tested the hypothesis that arsenic induces lasting muscle mitochondrial dysfunction and impairs metabolism. Compared to nonexposed controls, mice exposed to drinking water containing 100 μg/L arsenite for 5 weeks demonstrated impaired muscle function, mitochondrial myopathy, and altered oxygen consumption that were concomitant with increased mitochondrial fusion gene transcription. There were no differences in the levels of inorganic arsenic or its monomethyl and dimethyl metabolites between controls and exposed muscles, confirming that arsenic does not accumulate in muscle. Nevertheless, muscle progenitor cells isolated from exposed mice recapitulated the aberrant myofiber phenotype and were more resistant to oxidative stress, generated more reactive oxygen species, and displayed autophagic mitochondrial morphology, compared to cells isolated from nonexposed mice. These pathological changes from a possible maladaptive oxidative stress response provide insight into declines in muscle functioning caused by exposure to this common environmental contaminant.
Keywords: Free radicals; Metabolism; Mitochondria; Resistance to stress.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures

References
-
- Mukherjee SC, Rahman MM, Chowdhury UK, Sengupta MK, Lodh D, Chanda CR, Saha KC, Chakraborti D. Neuropathy in arsenic toxicity from groundwater arsenic contamination in West Bengal, India. J Environ Sci Health A Tox Hazard Subst Environ Eng. 2003;38:165–183. - PubMed
-
- Guha Mazumder DN. Chronic arsenic toxicity: clinical features, epidemiology, and treatment: experience in West Bengal. J.Environ.Sci Health Part A Tox.Hazard.Subst.Environ.Eng. 2003;38:141–163. - PubMed
-
- Doherty TJ. Invited review: Aging and sarcopenia. J Appl Physiol. 2003;95:1717–1727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

