Transarterial embolization therapies for the treatment of hepatocellular carcinoma: CEPO review and clinical recommendations
- PMID: 24961288
- PMCID: PMC4266441
- DOI: 10.1111/hpb.12273
Transarterial embolization therapies for the treatment of hepatocellular carcinoma: CEPO review and clinical recommendations
Abstract
Background: Hepatocellular carcinoma (HCC) is one of the most deadly cancers in the world and its incidence rate has consistently increased over the past 15 years in Canada. Although transarterial embolization therapies are palliative options commonly used for the treatment of HCC, their efficacy is still controversial. The objective of this guideline is to review the efficacy and safety of transarterial embolization therapies for the treatment of HCC and to develop evidence-based recommendations.
Method: A review of the scientific literature published up to October 2013 was performed. A total of 38 studies were included.
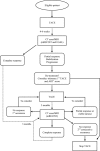
Recommendations: Considering the evidence available to date, the CEPO recommends the following: (i) transarterial chemoembolization therapy (TACE) be considered a standard of practice for the palliative treatment of HCC in eligible patients; (ii) drug-eluting beads (DEB)-TACE be considered an alternative and equivalent treatment to conventional TACE in terms of oncological efficacy (overall survival) and incidence of severe toxicities; (iii) the decision to treat with TACE or DEB-TACE be discussed in tumour boards; (iv) bland embolization (TAE) not be considered for the treatment of HCC; (v) radioembolization (TARE) not be considered outside of a clinical trial setting; and (vi) sorafenib combined with TACE not be considered outside of a clinical trial setting.
© 2014 International Hepato-Pancreato-Biliary Association.
Figures
References
-
- McGlynn KA, London WT. Epidemiology and natural history of hepatocellular carcinoma. Best Pract Res Clin Gastroenterol. 2005;19:3–23. - PubMed
-
- Comité consultatif de la Société canadienne du cancer. Statistiques canadiennes sur le cancer, 2013. Toronto, ON: Société canadienne du cancer; 2013.
-
- Lencioni R, Crocetti L. Local-regional treatment of hepatocellular carcinoma. Radiology. 2012;262:43–58. - PubMed
-
- Cook DJ, Guyatt GH, Laupacis A, Sackett DL. Rules of evidence and clinical recommendations on the use of antithrombotic agents. Chest. 1992;102(4 Suppl):305S–311S. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

