Macrophage-pathogen interactions in infectious diseases: new therapeutic insights from the zebrafish host model
- PMID: 24973749
- PMCID: PMC4073269
- DOI: 10.1242/dmm.015594
Macrophage-pathogen interactions in infectious diseases: new therapeutic insights from the zebrafish host model
Abstract
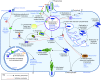
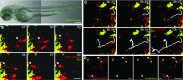
Studying macrophage biology in the context of a whole living organism provides unique possibilities to understand the contribution of this extremely dynamic cell subset in the reaction to infections, and has revealed the relevance of cellular and molecular processes that are fundamental to the cell-mediated innate immune response. In particular, various recently established zebrafish infectious disease models are contributing substantially to our understanding of the mechanisms by which different pathogens interact with macrophages and evade host innate immunity. Transgenic zebrafish lines with fluorescently labeled macrophages and other leukocyte populations enable non-invasive imaging at the optically transparent early life stages. Furthermore, there is a continuously expanding availability of vital reporters for subcellular compartments and for probing activation of immune defense mechanisms. These are powerful tools to visualize the activity of phagocytic cells in real time and shed light on the intriguing paradoxical roles of these cells in both limiting infection and supporting the dissemination of intracellular pathogens. This Review will discuss how several bacterial and fungal infection models in zebrafish embryos have led to new insights into the dynamic molecular and cellular mechanisms at play when pathogens encounter host macrophages. We also describe how these insights are inspiring novel therapeutic strategies for infectious disease treatment.
Keywords: Burkholderia; Candida; Host-directed therapy; Infectious disease; Innate immunity; Leukocyte biology; Mycobacterium; Salmonella; Shigella; Staphylococcus.
© 2014. Published by The Company of Biologists Ltd.
Figures

Similar articles
-
Neutrophils in host defense: new insights from zebrafish.J Leukoc Biol. 2015 Oct;98(4):523-37. doi: 10.1189/jlb.4MR1114-524R. Epub 2015 Feb 25. J Leukoc Biol. 2015. PMID: 25717145 Free PMC article. Review.
-
Modeling Infectious Diseases in the Context of a Developing Immune System.Curr Top Dev Biol. 2017;124:277-329. doi: 10.1016/bs.ctdb.2016.10.006. Epub 2016 Dec 19. Curr Top Dev Biol. 2017. PMID: 28335862 Review.
-
Zebrafish (Danio rerio) as a Model System to Investigate the Role of the Innate Immune Response in Human Infectious Diseases.Int J Mol Sci. 2024 Nov 8;25(22):12008. doi: 10.3390/ijms252212008. Int J Mol Sci. 2024. PMID: 39596075 Free PMC article. Review.
-
Infection of zebrafish embryos with intracellular bacterial pathogens.J Vis Exp. 2012 Mar 15;(61):3781. doi: 10.3791/3781. J Vis Exp. 2012. PMID: 22453760 Free PMC article.
-
Non-invasive imaging of disseminated candidiasis in zebrafish larvae.J Vis Exp. 2012 Jul 30;(65):4051. doi: 10.3791/4051. J Vis Exp. 2012. PMID: 22872032 Free PMC article.
Cited by
-
A new protocol for multispecies bacterial infections in zebrafish and their monitoring through automated image analysis.PLoS One. 2024 Aug 8;19(8):e0304827. doi: 10.1371/journal.pone.0304827. eCollection 2024. PLoS One. 2024. PMID: 39116043 Free PMC article.
-
Imaging Infection Across Scales of Size: From Whole Animals to Single Molecules.Annu Rev Microbiol. 2021 Oct 8;75:407-426. doi: 10.1146/annurev-micro-041521-121457. Epub 2021 Aug 3. Annu Rev Microbiol. 2021. PMID: 34343016 Free PMC article. Review.
-
The Diverse Cellular and Animal Models to Decipher the Physiopathological Traits of Mycobacterium abscessus Infection.Front Cell Infect Microbiol. 2017 Apr 4;7:100. doi: 10.3389/fcimb.2017.00100. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28421165 Free PMC article. Review.
-
Generation of mouse-zebrafish hematopoietic tissue chimeric embryos for hematopoiesis and host-pathogen interaction studies.Dis Model Mech. 2018 Nov 5;11(11):dmm034876. doi: 10.1242/dmm.034876. Dis Model Mech. 2018. PMID: 30266803 Free PMC article.
-
Phenotypic Plasticity Regulates Candida albicans Interactions and Virulence in the Vertebrate Host.Front Microbiol. 2016 May 26;7:780. doi: 10.3389/fmicb.2016.00780. eCollection 2016. Front Microbiol. 2016. PMID: 27303374 Free PMC article.
References
-
- Abdallah A. M., Gey van Pittius N. C., Champion P. A., Cox J., Luirink J., Vandenbroucke-Grauls C. M., Appelmelk B. J., Bitter W. (2007). Type VII secretion – mycobacteria show the way. Nat. Rev. Microbiol. 5, 883–891 - PubMed
-
- Agnoli K., Schwager S., Uehlinger S., Vergunst A., Viteri D. F., Nguyen D. T., Sokol P. A., Carlier A., Eberl L. (2012). Exposing the third chromosome of Burkholderia cepacia complex strains as a virulence plasmid. Mol. Microbiol. 83, 362–378 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

