A preliminary double-blind, placebo-controlled randomized study of baclofen effects in alcoholic smokers
- PMID: 24973894
- PMCID: PMC4278944
- DOI: 10.1007/s00213-014-3652-9
A preliminary double-blind, placebo-controlled randomized study of baclofen effects in alcoholic smokers
Erratum in
-
Erratum to: a preliminary double-blind, placebo-controlled randomized study of baclofen effects in alcoholic smokers.Psychopharmacology (Berl). 2015 May;232(9):1667. doi: 10.1007/s00213-015-3916-z. Psychopharmacology (Berl). 2015. PMID: 25804430 No abstract available.
Abstract
Rationale: There is presently no approved single treatment for dual alcohol and nicotine dependencies.
Objective: This pilot study investigated baclofen effects in alcoholic smokers.
Methods: This was a preliminary double-blind placebo-controlled randomized clinical study with 30 alcoholic smokers randomized to baclofen at 80 mg/day or placebo. A subgroup (n=18) participated in an alcohol cue-reactivity experiment.
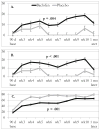
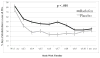
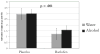
Results: Baclofen, compared with placebo, significantly decreased the percent days of abstinence from alcohol-tobacco co-use (p=0.004). Alcohol dependence severity moderated baclofen effects, with the higher severity group having the greater baclofen response (p<0.001). Although the percent days of alcohol-tobacco co-use declined in both groups, this decline was greater after placebo than baclofen (p<0.001). Secondary analyses on alcohol or tobacco use alone suggested that the increase in percent days of co-abstinence was driven by the medication differences on heavy drinking days and on percent days smoking. In the cue-reactivity substudy, baclofen slightly decreased alcohol urge (p=0.058) and significantly reduced salivation (p=0.001), but these effects were not related to cue type.
Conclusions: This study provides preliminary evidence suggesting a possible role of baclofen in the treatment of alcoholic smokers. However, the mixed results and the small sample require larger confirmatory studies.
Conflict of interest statement
Figures
References
-
- Addolorato G, Caputo F, Capristo E, Colombo G, Gessa GL, Gasbarrini G. Ability of baclofen in reducing alcohol craving and intake: II—preliminary clinical evidence. Alcohol Clin Exp Res. 2000;24:67–71. - PubMed
-
- Addolorato G, Caputo F, Capristo E, Domenicali M, Bernardi M, Janiri L, Agabio R, Colombo G, Gessa GL, Gasbarrini G. Baclofen efficacy in reducing alcohol craving and intake: a preliminary double-blind randomized controlled study. Alcohol Alcohol. 2002a;37:504–508. - PubMed
-
- Addolorato G, Caputo F, Capristo E, Janiri L, Bernardi M, Agabio R, Colombo G, Gessa GL, Gasbarrini G. Rapid suppression of alcohol withdrawal syndrome by baclofen. Am J Med. 2002b;112:226–229. - PubMed
-
- Addolorato G, Leggio L, Abenavoli L, Agabio R, Caputo F, Capristo E, Colombo G, Gessa GL, Gasbarrini G. Baclofen in the treatment of alcohol withdrawal syndrome: a comparative study vs diazepam. Am J Med. 2006;119(276):e213–e278. - PubMed
-
- Addolorato G, Leggio L, Ferrulli A, Cardone S, Vonghia L, Mirijello A, Abenavoli L, D’Angelo C, Caputo F, Zambon A, Haber PS, Gasbarrini G. Effectiveness and safety of baclofen for maintenance of alcohol abstinence in alcohol-dependent patients with liver cirrhosis: randomised, double-blind controlled study. Lancet. 2007;370:1915–1922. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

