iPSC-derived human mesenchymal stem cells improve myocardial strain of infarcted myocardium
- PMID: 24974908
- PMCID: PMC4190910
- DOI: 10.1111/jcmm.12351
iPSC-derived human mesenchymal stem cells improve myocardial strain of infarcted myocardium
Abstract
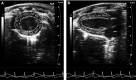
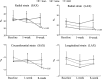
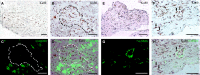
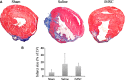
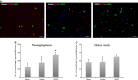
We investigated global and regional effects of myocardial transplantation of human induced pluripotent stem cell (iPSC)-derived mesenchymal stem cells (iMSCs) in infarcted myocardium. Acute myocardial infarction (MI) was induced by ligation of left coronary artery of severe combined immunodeficient mice before 2 × 10(5) iMSCs or cell-free saline were injected into peri-infarcted anterior free wall. Sham-operated animals received no injection. Global and regional myocardial function was assessed serially at 1-week and 8-week by segmental strain analysis by using two dimensional (2D) speckle tracking echocardiography. Early myocardial remodelling was observed at 1-week and persisted to 8-week with global contractility of ejection fraction and fractional area change in saline- (32.96 ± 14.23%; 21.50 ± 10.07%) and iMSC-injected (32.95 ± 10.31%; 21.00 ± 7.11%) groups significantly depressed as compared to sham control (51.17 ± 11.69%, P < 0.05; 34.86 ± 9.82%, P < 0.05). However, myocardial dilatation was observed in saline-injected animals (4.40 ± 0.62 mm, P < 0.05), but not iMSCs (4.29 ± 0.57 mm), when compared to sham control (3.74 ± 0.32 mm). Furthermore, strain analysis showed significant improved basal anterior wall strain (28.86 ± 8.16%, P < 0.05) in the iMSC group, but not saline-injected (15.81 ± 13.92%), when compared to sham control (22.18 ± 4.13%). This was corroborated by multi-segments deterioration of radial strain only in saline-injected (21.50 ± 5.31%, P < 0.05), but not iMSC (25.67 ± 12.53%), when compared to sham control (34.88 ± 5.77%). Improvements of the myocardial strain coincided with the presence of interconnecting telocytes in interstitial space of the infarcted anterior segment of the heart. Our results show that localized injection of iMSCs alleviates ventricular remodelling, sustains global and regional myocardial strain by paracrine-driven effect on neoangiogenesis and myocardial deformation/compliance via parenchymal and interstitial cell interactions in the infarcted myocardium.
Keywords: cell therapy; myocardial compliance; myocardial strain; telocytes; tissue deformation.
© 2014 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures


References
-
- Miettinen JA, Ylitalo K, Hedberg P, et al. Determinants of functional recovery after myocardial infarction of patients treated with bone marrow-derived stem cells after thrombolytic therapy. Heart. 2010;96:362–7. - PubMed
-
- Chen SL, Fang WW, Qian J, et al. Improvement of cardiac function after transplantation of autologous bone marrow mesenchymal stem cells in patients with acute myocardial infarction. Chin Med J (Engl) 2004;117:1443–8. - PubMed
-
- Mehta A, Shim W. Cardiac stem cell therapy: stemness or commitment? Cell Transplant. 2013;22:1–14. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

