Hepatitis B virus reactivation in patients with hepatocellular carcinoma undergoing anti-cancer therapy
- PMID: 24976705
- PMCID: PMC4069296
- DOI: 10.3748/wjg.v20.i24.7675
Hepatitis B virus reactivation in patients with hepatocellular carcinoma undergoing anti-cancer therapy
Abstract
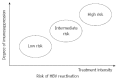
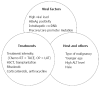
Patients with hepatocellular carcinoma (HCC) often experience hepatic morbidity. Hepatitis B virus (HBV) reactivation is well documented as a serious hepatic morbidity during anti-cancer therapy. Reported rates of HBV reactivation in chronic carriers with HCC undergoing chemotherapy range from 4%-67%. Apart from chemotherapy, HBV reactivation has been increasingly identified in settings of hepatectomy and local ablation therapies. The rates of HBV reactivation vary with different levels of immunosuppression and depend on treatment, viral factors, and patient characteristics. The principal concern relating to reactivation is that a substantial proportion of patients with reactivation suffer from liver dysfunction during therapy, which often leads to disruption of planned, potentially life-prolonging treatments, adversely affecting the patients' final outcome. The first step in the management of HBV reactivation is identification of patients at risk of reactivation by testing for HBV serology prior to commencing anti-cancer therapy. Although it is a serious complication, HBV reactivation is preventable with prophylactic anti-HBV drugs. Multiple publications have shown the benefit of prophylactic or preemptive antiviral therapy in this setting and justified such an approach before the start of therapy. Given the tumors and underlying cirrhosis, long-term use of antivirals with high potency and low risk of resistance is recommended in patients with HCC. This topic review will summarize the epidemiology, pathogenesis, and clinical issues related to HBV reactivation in HCC patients, and will discuss proper management against HBV reactivation during anti-cancer therapy for HCC.
Keywords: Hepatic morbidity; Hepatitis B virus; Hepatocellular carcinoma; Immunosuppression; Virus activation.
Figures
References
-
- Hoofnagle JH. Reactivation of hepatitis B. Hepatology. 2009;49:S156–S165. - PubMed
-
- Perrillo RP. Acute flares in chronic hepatitis B: the natural and unnatural history of an immunologically mediated liver disease. Gastroenterology. 2001;120:1009–1022. - PubMed
-
- Yeo W, Johnson PJ. Diagnosis, prevention and management of hepatitis B virus reactivation during anticancer therapy. Hepatology. 2006;43:209–220. - PubMed
-
- Kusumoto S, Tanaka Y, Mizokami M, Ueda R. Reactivation of hepatitis B virus following systemic chemotherapy for malignant lymphoma. Int J Hematol. 2009;90:13–23. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

