Evaluation of developmental toxicity of propylthiouracil and methimazole
- PMID: 24980470
- PMCID: PMC4295064
- DOI: 10.1002/bdrb.21113
Evaluation of developmental toxicity of propylthiouracil and methimazole
Abstract
Background: Propylthiouracil (PTU) and methimazole (MMI) are antithyroid drugs used to treat hyperthyroidism. Despite the widespread use of PTU and MMI during pregnancy, modest clinical data and less animal data are available on the teratogenic potential of these drugs.
Methods: We evaluated the teratogenicity of in utero exposure to PTU or MMI in mice and rats. First, pregnant C57Bl/6 mice were treated daily with PTU (10 or 100 mg/kg), MMI (2 or 20 mg/kg), or vehicle from gestation day (GD) 6 to 16. GD 18 fetuses were evaluated for gross and histopathological abnormalities. Next, pregnant Sprague-Dawley rats were treated daily with PTU (50 or 100 mg/kg), MMI (10 or 20 mg/kg), or vehicle from GD 6 to 19, followed by evaluation for gross and histopathological abnormalities at GD 20.
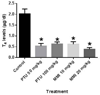
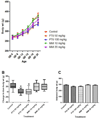
Results: In mice treated with PTU or MMI, no significant histopathological abnormalities or external gross malformations, and no adverse effects on placental weight, litter size, resorption rates, or fetal weight were observed at GD 18. In rats, no adverse effects on litter size, placental weights, or maternal body weights were observed with either PTU or MMI treatment. PTU treatment (50 and 100 mg/kg) and MMI (10 mg/kg) treatment resulted in a decrease in crown-rump length in rat fetuses but no external gross malformations or histopathological abnormalities were observed.
Conclusion: We did not observe either gross external malformations or histopathological malformations in mice or rats treated long-term with high doses of PTU or MMI during pregnancy.
Keywords: antithyroid drugs; developmental toxicity; methimazole; propylthiouracil.
© 2014 Wiley Periodicals, Inc.
Figures



Similar articles
-
Propylthiouracil is teratogenic in murine embryos.PLoS One. 2012;7(4):e35213. doi: 10.1371/journal.pone.0035213. Epub 2012 Apr 18. PLoS One. 2012. PMID: 22529993 Free PMC article.
-
Embryonic exposure to propylthiouracil disrupts left-right patterning in Xenopus embryos.FASEB J. 2013 Feb;27(2):684-91. doi: 10.1096/fj.12-218073. Epub 2012 Nov 12. FASEB J. 2013. PMID: 23150524 Free PMC article.
-
The safety of methimazole and propylthiouracil in pregnancy: a systematic review.J Obstet Gynaecol Can. 2012 Nov;34(11):1077-1086. doi: 10.1016/S1701-2163(16)35438-X. J Obstet Gynaecol Can. 2012. PMID: 23231846
-
Birth defects after early pregnancy use of antithyroid drugs: a Danish nationwide study.J Clin Endocrinol Metab. 2013 Nov;98(11):4373-81. doi: 10.1210/jc.2013-2831. Epub 2013 Oct 22. J Clin Endocrinol Metab. 2013. PMID: 24151287
-
Toxicological considerations for antithyroid drugs in children.Expert Opin Drug Metab Toxicol. 2011 Apr;7(4):399-410. doi: 10.1517/17425255.2011.557068. Epub 2011 Feb 17. Expert Opin Drug Metab Toxicol. 2011. PMID: 21323607 Review.
Cited by
-
Effects of Maternal Hypothyroidism on the Pubertal Development in Female Rat Offspring.Dev Reprod. 2021 Jun;25(2):83-91. doi: 10.12717/DR.2021.25.2.83. Epub 2021 Jun 30. Dev Reprod. 2021. PMID: 34386643 Free PMC article.
-
Repositioning the existing drugs for neuroinflammation: a fusion of computational approach and biological validation to counter the Parkinson's disease progression.Mol Divers. 2024 Oct;28(5):2759-2770. doi: 10.1007/s11030-023-10708-5. Epub 2023 Aug 4. Mol Divers. 2024. PMID: 37542020
-
Treatment of Graves' hyperthyroidism with thionamides: a position paper on indications and safety in pregnancy.J Endocrinol Invest. 2020 Feb;43(2):257-265. doi: 10.1007/s40618-019-01148-w. Epub 2019 Nov 29. J Endocrinol Invest. 2020. PMID: 31784912 Review.
-
Compensatory mechanisms in response to induced hypothyroidism in the late gestation pig fetus†.Biol Reprod. 2023 May 10;108(5):731-743. doi: 10.1093/biolre/ioad024. Biol Reprod. 2023. PMID: 36811850 Free PMC article.
-
Exposure to the UV Filter Octyl Methoxy Cinnamate in the Postnatal Period Induces Thyroid Dysregulation and Perturbs the Immune System of Mice.Front Endocrinol (Lausanne). 2020 Jan 31;10:943. doi: 10.3389/fendo.2019.00943. eCollection 2019. Front Endocrinol (Lausanne). 2020. PMID: 32082254 Free PMC article.
References
-
- Abalovich M, Amino N, Barbour LA, Cobin RH, De Groot LJ, Glinoer D, Mandel SJ, Stagnaro-Green A. Management of thyroid dysfunction during pregnancy and postpartum: An endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2007;92(8 Suppl):S1–S47. - PubMed
-
- Andersen SL, Olsen J, Wu CS, Laurberg P. Birth defects after early pregnancy use of antithyroid drugs: A danish nationwide study. J Clin Endocrinol Metab. 2013;98(11):4373–4381. - PubMed
-
- Bahn RS, Burch HS, Cooper DS, Garber JR, Greenlee CM, Klein IL, Laurberg P, McDougall IR, Rivkees SA, Ross D, Sosa JA, Stan MN. The role of propylthiouracil in the management of graves' disease in adults: Report of a meeting jointly sponsored by the american thyroid association and the food and drug administration. Thyroid. 2009;19(7):673–674. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

