The evolving role of chemical synthesis in antibacterial drug discovery
- PMID: 24990531
- PMCID: PMC4536949
- DOI: 10.1002/anie.201310843
The evolving role of chemical synthesis in antibacterial drug discovery
Abstract
The discovery and implementation of antibiotics in the early twentieth century transformed human health and wellbeing. Chemical synthesis enabled the development of the first antibacterial substances, organoarsenicals and sulfa drugs, but these were soon outshone by a host of more powerful and vastly more complex antibiotics from nature: penicillin, streptomycin, tetracycline, and erythromycin, among others. These primary defences are now significantly less effective as an unavoidable consequence of rapid evolution of resistance within pathogenic bacteria, made worse by widespread misuse of antibiotics. For decades medicinal chemists replenished the arsenal of antibiotics by semisynthetic and to a lesser degree fully synthetic routes, but economic factors have led to a subsidence of this effort, which places society on the precipice of a disaster. We believe that the strategic application of modern chemical synthesis to antibacterial drug discovery must play a critical role if a crisis of global proportions is to be averted.
Keywords: antibiotics; chemical synthesis; drug discovery; semisynthesis.
© 2014 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures








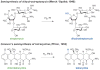




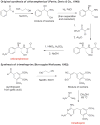
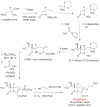
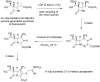
References
-
- World Economic Forum. Global Risks Report 2013. [accessed October, 2013]; http://www.weforum.org/reports/global-risks-2013-eighth-edition.
-
- CDC's Antibiotic Resistance Threats in the United States, 2013. [accessed October, 2013]; http://www.cdc.gov/drugresistance/threat-report-2013/pdf/ar-threats-2013....
-
- The Center for Disease Dynamics, Economics & Policy, Resistance Map. [accessed October, 2013]; http://www.cddep.org/map.
-
-
a) For an example of carbapenem resistance conferred by New Delhi metallo-β-lactamases (NDMs, also known as plasmid-encoding, carbapenem-resistant metallo-β-lactamases, PCMs), see: Kumarasamy KK, Toleman MA, Walsh TR, Bagaria J, Butt F, Balakrishnan R, Chaudhary U, Doumith M, Giske CG, Irfan S, Krishnan P, Kumar AV, Maharjan S, Mushtaq S, Noorie T, Paterson DL, Pearson A, Perry C, Pike R, Rao B, Ray U, Sarma JB, Sharma M, Sheridan E, Thirunarayan MA, Turton J, Upadhyay S, Warner M, Welfare W, Livermore DM, Woodford N. Lancet Infect Dis. 2010;10:597–602.; b) see also: Moellering RC., Jr N Engl J Med. 2010;363:2377–2379.; c) For an example of a Klebsiella pneumoniae carbapenemase, see: Snitkin ES, Zelazny AM, Thomas PJ, Stock F, Henderson DK, Palmore TN, Segre JA N. C. S. P. Group. Sci Transl Med. 2012;4:148ra116.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

