Functional specialization in rat occipital and temporal visual cortex
- PMID: 24990566
- PMCID: PMC4200005
- DOI: 10.1152/jn.00737.2013
Functional specialization in rat occipital and temporal visual cortex
Abstract
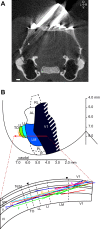
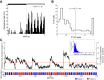
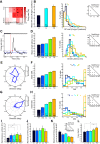
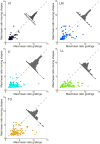
Recent studies have revealed a surprising degree of functional specialization in rodent visual cortex. Anatomically, suggestions have been made about the existence of hierarchical pathways with similarities to the ventral and dorsal pathways in primates. Here we aimed to characterize some important functional properties in part of the supposed "ventral" pathway in rats. We investigated the functional properties along a progression of five visual areas in awake rats, from primary visual cortex (V1) over lateromedial (LM), latero-intermediate (LI), and laterolateral (LL) areas up to the newly found lateral occipito-temporal cortex (TO). Response latency increased >20 ms from areas V1/LM/LI to areas LL and TO. Orientation and direction selectivity for the used grating patterns increased gradually from V1 to TO. Overall responsiveness and selectivity to shape stimuli decreased from V1 to TO and was increasingly dependent upon shape motion. Neural similarity for shapes could be accounted for by a simple computational model in V1, but not in the other areas. Across areas, we find a gradual change in which stimulus pairs are most discriminable. Finally, tolerance to position changes increased toward TO. These findings provide unique information about possible commonalities and differences between rodents and primates in hierarchical cortical processing.
Keywords: high-level vision; population coding; position tolerance; rodent research; single-unit recordings.
Copyright © 2014 the American Physiological Society.
Figures





Similar articles
-
Nonlinear Processing of Shape Information in Rat Lateral Extrastriate Cortex.J Neurosci. 2019 Feb 27;39(9):1649-1670. doi: 10.1523/JNEUROSCI.1938-18.2018. Epub 2019 Jan 7. J Neurosci. 2019. PMID: 30617210 Free PMC article.
-
Neural discriminability in rat lateral extrastriate cortex and deep but not superficial primary visual cortex correlates with shape discriminability.Front Neural Circuits. 2015 May 20;9:24. doi: 10.3389/fncir.2015.00024. eCollection 2015. Front Neural Circuits. 2015. PMID: 26041999 Free PMC article.
-
Hierarchical stimulus processing in rodent primary and lateral visual cortex as assessed through neuronal selectivity and repetition suppression.J Neurophysiol. 2018 Sep 1;120(3):926-941. doi: 10.1152/jn.00673.2017. Epub 2018 May 9. J Neurophysiol. 2018. PMID: 29742022 Free PMC article.
-
Functional cell classes and functional architecture in the early visual system of a highly visual rodent.Prog Brain Res. 2005;149:127-45. doi: 10.1016/S0079-6123(05)49010-X. Prog Brain Res. 2005. PMID: 16226581 Review.
-
Human cortical areas underlying the perception of optic flow: brain imaging studies.Int Rev Neurobiol. 2000;44:269-92. doi: 10.1016/s0074-7742(08)60746-1. Int Rev Neurobiol. 2000. PMID: 10605650 Review.
Cited by
-
Emergence of transformation-tolerant representations of visual objects in rat lateral extrastriate cortex.Elife. 2017 Apr 11;6:e22794. doi: 10.7554/eLife.22794. Elife. 2017. PMID: 28395730 Free PMC article.
-
Hunger-Dependent Enhancement of Food Cue Responses in Mouse Postrhinal Cortex and Lateral Amygdala.Neuron. 2016 Sep 7;91(5):1154-1169. doi: 10.1016/j.neuron.2016.07.032. Epub 2016 Aug 11. Neuron. 2016. PMID: 27523426 Free PMC article.
-
Nonlinear Processing of Shape Information in Rat Lateral Extrastriate Cortex.J Neurosci. 2019 Feb 27;39(9):1649-1670. doi: 10.1523/JNEUROSCI.1938-18.2018. Epub 2019 Jan 7. J Neurosci. 2019. PMID: 30617210 Free PMC article.
-
Efficient processing of natural scenes in visual cortex.Front Cell Neurosci. 2022 Dec 5;16:1006703. doi: 10.3389/fncel.2022.1006703. eCollection 2022. Front Cell Neurosci. 2022. PMID: 36545653 Free PMC article. Review.
-
Unraveling the complexity of rat object vision requires a full convolutional network and beyond.Patterns (N Y). 2025 Jan 17;6(2):101149. doi: 10.1016/j.patter.2024.101149. eCollection 2025 Feb 14. Patterns (N Y). 2025. PMID: 40041851 Free PMC article.
References
-
- Brainard DH. The Psychophysics Toolbox. Spat Vis 10: 433–436, 1997 - PubMed
-
- Braje WL, Kersten D, Tarr MJ, Troje NF. Illumination effects in face recognition. Psychobiology 26 371–380, 1998
-
- Chelazzi L, Rossi F, Tempia F, Ghirardi M, Strata P. Saccadic eye movements and gaze holding in the head-restrained pigmented rat. Eur J Neurosci 1: 639–646, 1989 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

