Carbohydrate PEGylation, an approach to improve pharmacological potency
- PMID: 24991298
- PMCID: PMC4077506
- DOI: 10.3762/bjoc.10.147
Carbohydrate PEGylation, an approach to improve pharmacological potency
Abstract
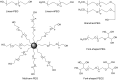
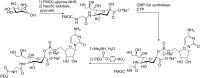
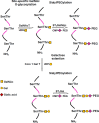
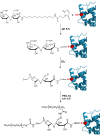
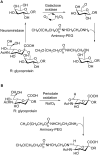
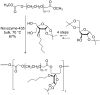
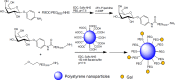
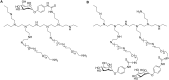
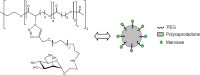
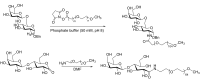
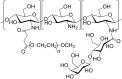
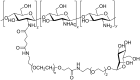
Conjugation with polyethylene glycol (PEG), known as PEGylation, has been widely used to improve the bioavailability of proteins and low molecular weight drugs. The covalent conjugation of PEG to the carbohydrate moiety of a protein has been mainly used to enhance the pharmacokinetic properties of the attached protein while yielding a more defined product. Thus, glycoPEGylation was successfully applied to the introduction of a PEGylated sialic acid to a preexisting or enzymatically linked glycan in a protein. Carbohydrates are now recognized as playing an important role in host-pathogen interactions in protozoal, bacterial and viral infections and are consequently candidates for chemotherapy. The short in vivo half-life of low molecular weight glycans hampered their use but methods for the covalent attachment of PEG have been less exploited. In this review, information on the preparation and application of PEG-carbohydrates, in particular multiarm PEGylation, is presented.
Keywords: bioavailability; carbohydrates; conjugates; glycoPEGylation; multivalent PEGylation; multivalent glycosystems.
Figures

Similar articles
-
GlycoPEGylation of recombinant therapeutic proteins produced in Escherichia coli.Glycobiology. 2006 Sep;16(9):833-43. doi: 10.1093/glycob/cwl004. Epub 2006 May 22. Glycobiology. 2006. PMID: 16717104
-
The impact of PEGylation on biological therapies.BioDrugs. 2008;22(5):315-29. doi: 10.2165/00063030-200822050-00004. BioDrugs. 2008. PMID: 18778113 Review.
-
Chemical and enzymatic site specific PEGylation of hGH.Bioconjug Chem. 2013 Mar 20;24(3):456-63. doi: 10.1021/bc300594y. Epub 2013 Mar 4. Bioconjug Chem. 2013. PMID: 23432141
-
Chemical and Enzymatic Site Specific PEGylation of hGH: The Stability and in vivo Activity of PEG-N-Terminal-hGH and PEG-Gln141-hGH Conjugates.Macromol Biosci. 2016 Jan;16(1):50-6. doi: 10.1002/mabi.201500282. Epub 2015 Sep 9. Macromol Biosci. 2016. PMID: 26350165
-
From Synthesis to Characterization of Site-Selective PEGylated Proteins.Front Pharmacol. 2019 Dec 18;10:1450. doi: 10.3389/fphar.2019.01450. eCollection 2019. Front Pharmacol. 2019. PMID: 31920645 Free PMC article. Review.
Cited by
-
Synthesis and facile end-group quantification of functionalized PEG azides.J Polym Sci A Polym Chem. 2016 Sep 15;54(18):2888-2895. doi: 10.1002/pola.28174. Epub 2016 Jun 14. J Polym Sci A Polym Chem. 2016. PMID: 27840557 Free PMC article.
-
An EPR Strategy for Bio-responsive Fluorescence Guided Surgery with Simulation of the Benefit for Imaging.Theranostics. 2020 Feb 10;10(7):3064-3082. doi: 10.7150/thno.42702. eCollection 2020. Theranostics. 2020. PMID: 32194855 Free PMC article.
-
Thiodigalactoside-Bovine Serum Albumin Conjugates as High-Potency Inhibitors of Galectin-3: An Outstanding Example of Multivalent Presentation of Small Molecule Inhibitors.Bioconjug Chem. 2018 Apr 18;29(4):1266-1275. doi: 10.1021/acs.bioconjchem.8b00047. Epub 2018 Mar 7. Bioconjug Chem. 2018. PMID: 29474087 Free PMC article.
-
Investigational chemotherapy and novel pharmacokinetic mechanisms for the treatment of breast cancer brain metastases.Pharmacol Res. 2018 Jun;132:47-68. doi: 10.1016/j.phrs.2018.03.021. Epub 2018 Mar 28. Pharmacol Res. 2018. PMID: 29604436 Free PMC article. Review.
-
Partial Surface Modification of Low Generation Polyamidoamine Dendrimers: Gaining Insight into their Potential for Improved Carboplatin Delivery.Biomolecules. 2019 Jun 2;9(6):214. doi: 10.3390/biom9060214. Biomolecules. 2019. PMID: 31159469 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
