The compromise of macrophage functions by hyperoxia is attenuated by ethacrynic acid via inhibition of NF-κB-mediated release of high-mobility group box-1
- PMID: 24992505
- PMCID: PMC4370245
- DOI: 10.1165/rcmb.2013-0544OC
The compromise of macrophage functions by hyperoxia is attenuated by ethacrynic acid via inhibition of NF-κB-mediated release of high-mobility group box-1
Abstract
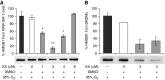
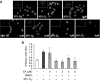
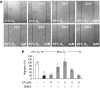
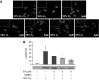
The prolonged exposure to hyperoxia can compromise macrophage functions and contribute to the development of ventilator-associated pneumonia. High levels of extracellular high-mobility group box-1 (HMGB1) in the airways of mice exposed to hyperoxia can directly cause macrophage dysfunction. Hence, inhibition of the release of nuclear HMGB1 into the extracellular milieu may help to maintain macrophage functions under hyperoxic conditions. The present study investigates whether ethacrynic acid (EA) affects hyperoxia-induced HMGB1 release from macrophages and improves their functions. Macrophage-like RAW 264.7 cells and bone marrow-derived macrophages were exposed to different concentrations of EA for 24 hours in the presence of 95% O2. EA significantly decreased the accumulation of extracellular HMGB1 in cultured media. Importantly, the phagocytic activity and migration capability of macrophages were significantly enhanced in EA-treated cells. Interestingly, hyperoxia-induced NF-κB activation was also inhibited in these cells. To determine whether NF-κB plays a role in hyperoxia-induced HMGB1 release, BAY 11-7082, an inhibitor of NF-κB activation, was used. Similar to EA, BAY 11-7082 significantly inhibited the accumulation of extracellular HMGB1 and improved hyperoxia-compromised macrophage migration and phagocytic activity. Furthermore, 24-hour hyperoxic exposure of macrophages caused hyperacetylation of HMGB1 and its subsequent cytoplasmic translocation and release, which were inhibited by EA and BAY 11-7082. Together, these results suggest that EA enhances hyperoxia-compromised macrophage functions by inhibiting HMGB1 hyperacetylation and its release from macrophages, possibly through attenuation of the NF-κB activation. Therefore, the activation of NF-κB could be one of the underlying mechanisms that mediate hyperoxia-compromised macrophage functions.
Keywords: NF-κB; high-mobility group box-1; hyperoxia; macrophage; phagocytosis.
Figures



Similar articles
-
The phase 2 enzyme inducers ethacrynic acid, DL-sulforaphane, and oltipraz inhibit lipopolysaccharide-induced high-mobility group box 1 secretion by RAW 264.7 cells.J Pharmacol Exp Ther. 2006 Mar;316(3):1070-9. doi: 10.1124/jpet.105.092841. Epub 2005 Dec 28. J Pharmacol Exp Ther. 2006. PMID: 16382025
-
The α7 nicotinic acetylcholine receptor agonist GTS-21 improves bacterial clearance in mice by restoring hyperoxia-compromised macrophage function.Mol Med. 2014 Jun 19;20(1):238-47. doi: 10.2119/molmed.2013.00086. Mol Med. 2014. Retraction in: Mol Med. 2020 Dec 30;26(1):132. doi: 10.1186/s10020-020-00265-0. PMID: 24664237 Free PMC article. Retracted.
-
The α7 nicotinic acetylcholine receptor agonist GTS-21 improves bacterial clearance in mice by restoring hyperoxia-compromised macrophage function.Mol Med. 2020 Oct 30;26(1):98. doi: 10.1186/s10020-020-00224-9. Mol Med. 2020. PMID: 33126860 Free PMC article.
-
A dangerous duo in adipose tissue: high-mobility group box 1 protein and macrophages.Yale J Biol Med. 2014 Jun 6;87(2):127-33. eCollection 2014 Jun. Yale J Biol Med. 2014. PMID: 24910558 Free PMC article. Review.
-
Manipulation of gene expression by oxygen: a primer from bedside to bench.Pediatr Res. 2009 Jul;66(1):3-10. doi: 10.1203/PDR.0b013e3181a2c184. Pediatr Res. 2009. PMID: 19287338 Free PMC article. Review.
Cited by
-
Affect of Early Life Oxygen Exposure on Proper Lung Development and Response to Respiratory Viral Infections.Front Med (Lausanne). 2015 Aug 10;2:55. doi: 10.3389/fmed.2015.00055. eCollection 2015. Front Med (Lausanne). 2015. PMID: 26322310 Free PMC article. Review.
-
The Positive Allosteric Modulation of alpha7-Nicotinic Cholinergic Receptors by GAT107 Increases Bacterial Lung Clearance in Hyperoxic Mice by Decreasing Oxidative Stress in Macrophages.Antioxidants (Basel). 2021 Jan 19;10(1):135. doi: 10.3390/antiox10010135. Antioxidants (Basel). 2021. PMID: 33477969 Free PMC article.
-
Infection-induced innate antimicrobial response disorders: from signaling pathways and their modulation to selected biomarkers.Cent Eur J Immunol. 2020;45(1):104-116. doi: 10.5114/ceji.2020.94712. Cent Eur J Immunol. 2020. PMID: 32425688 Free PMC article. Review.
-
High Mobility Group Box 1 (HMGB1): Molecular Signaling and Potential Therapeutic Strategies.Cells. 2024 Nov 23;13(23):1946. doi: 10.3390/cells13231946. Cells. 2024. PMID: 39682695 Free PMC article. Review.
-
Ethyl pyruvate attenuates murine allergic rhinitis partly by decreasing high mobility group box 1 release.Exp Biol Med (Maywood). 2015 Nov;240(11):1490-9. doi: 10.1177/1535370214566563. Epub 2015 Feb 13. Exp Biol Med (Maywood). 2015. PMID: 25681468 Free PMC article.
References
-
- Make BJ, Hill NS, Goldberg AI, Bach JR, Criner GJ, Dunne PE, et al. Mechanical ventilation beyond the intensive care unit: report of a consensus conference of the American College of Chest Physicians. Chest. 1998;113(5) suppl:289S–344S. - PubMed
-
- Davis KA. Ventilator-associated pneumonia: a review. J Intensive Care Med. 2006;21:211–226. - PubMed
-
- Craven DE, Kunches LM, Kilinsky V, Lichtenberg DA, Make BJ, McCabe WR. Risk factors for pneumonia and fatality in patients receiving continuous mechanical ventilation. Am Rev Respir Dis. 1986;133:792–796. - PubMed
-
- Rello J, Lorente C, Diaz E, Bodi M, Boque C, Sandiumenge A, et al. Incidence, etiology, and outcome of nosocomial pneumonia in ICU patients requiring percutaneous tracheotomy for mechanical ventilation. Chest. 2003;124:2239–2243. - PubMed
-
- Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med. 2002;165:867–903. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

