Chronic hypoxia limits H2O2-induced inhibition of ASIC1-dependent store-operated calcium entry in pulmonary arterial smooth muscle
- PMID: 24993130
- PMCID: PMC4154254
- DOI: 10.1152/ajplung.00095.2014
Chronic hypoxia limits H2O2-induced inhibition of ASIC1-dependent store-operated calcium entry in pulmonary arterial smooth muscle
Abstract
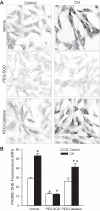
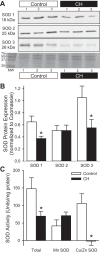
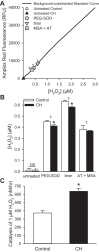
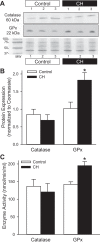
Our laboratory shows that acid-sensing ion channel 1 (ASIC1) contributes to the development of hypoxic pulmonary hypertension by augmenting store-operated Ca(2+) entry (SOCE) that is associated with enhanced agonist-induced vasoconstriction and arterial remodeling. However, this enhanced Ca(2+) influx following chronic hypoxia (CH) is not dependent on an increased ASIC1 protein expression in pulmonary arterial smooth muscle cells (PASMC). It is well documented that hypoxic pulmonary hypertension is associated with changes in redox potential and reactive oxygen species homeostasis. ASIC1 is a redox-sensitive channel showing increased activity in response to reducing agents, representing an alternative mechanism of regulation. We hypothesize that the enhanced SOCE following CH results from removal of an inhibitory effect of hydrogen peroxide (H2O2) on ASIC1. We found that CH increased PASMC superoxide (O2 (·-)) and decreased rat pulmonary arterial H2O2 levels. This decrease in H2O2 is a result of decreased Cu/Zn superoxide dismutase expression and activity, as well as increased glutathione peroxidase (GPx) expression and activity following CH. Whereas H2O2 inhibited ASIC1-dependent SOCE in PASMC from control and CH animals, addition of catalase augmented ASIC1-mediated SOCE in PASMC from control rats but had no further effect in PASMC from CH rats. These data suggest that, under control conditions, H2O2 inhibits ASIC1-dependent SOCE. Furthermore, H2O2 levels are decreased following CH as a result of diminished dismutation of O2 (·-) and increased H2O2 catalysis through GPx-1, leading to augmented ASIC1-dependent SOCE.
Keywords: catalase; degenerin/epithelial sodium channel; di-(4-carboxybenzyl) hyponitrite-1; glutathione peroxidase; pulmonary hypertension; superoxide dismutase.
Copyright © 2014 the American Physiological Society.
Figures





Similar articles
-
Loss of acid-sensing ion channel 2 enhances pulmonary vascular resistance and hypoxic pulmonary hypertension.J Appl Physiol (1985). 2019 Aug 1;127(2):393-407. doi: 10.1152/japplphysiol.00894.2018. Epub 2019 Jun 6. J Appl Physiol (1985). 2019. PMID: 31169471 Free PMC article.
-
Role of ASIC1 in the development of chronic hypoxia-induced pulmonary hypertension.Am J Physiol Heart Circ Physiol. 2014 Jan 1;306(1):H41-52. doi: 10.1152/ajpheart.00269.2013. Epub 2013 Nov 1. Am J Physiol Heart Circ Physiol. 2014. PMID: 24186095 Free PMC article.
-
Acid-sensing ion channel 1 contributes to pulmonary arterial smooth muscle cell depolarization following hypoxic pulmonary hypertension.J Physiol. 2021 Nov;599(21):4749-4762. doi: 10.1113/JP282231. Epub 2021 Sep 25. J Physiol. 2021. PMID: 34487355 Free PMC article.
-
PICK1/calcineurin suppress ASIC1-mediated Ca2+ entry in rat pulmonary arterial smooth muscle cells.Am J Physiol Cell Physiol. 2016 Mar 1;310(5):C390-400. doi: 10.1152/ajpcell.00091.2015. Epub 2015 Dec 23. Am J Physiol Cell Physiol. 2016. PMID: 26702130 Free PMC article.
-
Smooth muscle acid-sensing ion channel 1: pathophysiological implication in hypoxic pulmonary hypertension.Exp Physiol. 2015 Feb 1;100(2):111-20. doi: 10.1113/expphysiol.2014.081612. Epub 2015 Jan 14. Exp Physiol. 2015. PMID: 25398716 Review.
Cited by
-
Augmented Pulmonary Vasoconstrictor Reactivity after Chronic Hypoxia Requires Src Kinase and Epidermal Growth Factor Receptor Signaling.Am J Respir Cell Mol Biol. 2020 Jan;62(1):61-73. doi: 10.1165/rcmb.2018-0106OC. Am J Respir Cell Mol Biol. 2020. PMID: 31264901 Free PMC article.
-
Intermittent Hypoxia Augments Pulmonary Vasoconstrictor Reactivity through PKCβ/Mitochondrial Oxidant Signaling.Am J Respir Cell Mol Biol. 2020 Jun;62(6):732-746. doi: 10.1165/rcmb.2019-0351OC. Am J Respir Cell Mol Biol. 2020. PMID: 32048876 Free PMC article.
-
Isolation, culture and identification of pulmonary arterial smooth muscle cells from rat distal pulmonary arteries.Cytotechnology. 2017 Oct;69(5):831-840. doi: 10.1007/s10616-017-0081-8. Epub 2017 Mar 21. Cytotechnology. 2017. PMID: 28321780 Free PMC article.
-
Loss of acid-sensing ion channel 2 enhances pulmonary vascular resistance and hypoxic pulmonary hypertension.J Appl Physiol (1985). 2019 Aug 1;127(2):393-407. doi: 10.1152/japplphysiol.00894.2018. Epub 2019 Jun 6. J Appl Physiol (1985). 2019. PMID: 31169471 Free PMC article.
-
PKCβ and reactive oxygen species mediate enhanced pulmonary vasoconstrictor reactivity following chronic hypoxia in neonatal rats.Am J Physiol Heart Circ Physiol. 2020 Feb 1;318(2):H470-H483. doi: 10.1152/ajpheart.00629.2019. Epub 2020 Jan 10. Am J Physiol Heart Circ Physiol. 2020. PMID: 31922892 Free PMC article.
References
-
- Andrey F, Tsintsadze T, Volkova T, Lozovaya N, Krishtal O. Acid sensing ionic channels: Modulation by redox reagents. Biochim Biophys Acta 1745: 1–6, 2005 - PubMed
-
- Archer SL, Huang J, Henry T, Peterson D, Weir EK. A redox-based O2 sensor in rat pulmonary vasculature. Circ Res 73: 1100–1112, 1993 - PubMed
-
- Archer SL, Marsboom G, Kim GH, Zhang HJ, Toth PT, Svensson EC, Dyck JR, Gomberg-Maitland M, Thebaud B, Husain AN, Cipriani N, Rehman J. Epigenetic attenuation of mitochondrial superoxide dismutase 2 in pulmonary arterial hypertension: a basis for excessive cell proliferation and a new therapeutic target. Circulation 121: 2661–2671, 2010 - PMC - PubMed
-
- Benarroch EE. Acid-sensing cation channels: structure, function, and pathophysiologic implications. Neurology 82: 628–635, 2014 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

