Acceptability of the Nestorone®/ethinyl estradiol contraceptive vaginal ring: development of a model; implications for introduction
- PMID: 24993487
- PMCID: PMC4589411
- DOI: 10.1016/j.contraception.2014.05.015
Acceptability of the Nestorone®/ethinyl estradiol contraceptive vaginal ring: development of a model; implications for introduction
Abstract
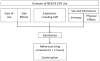
Objectives: Develop and test a theoretical acceptability model for the Nestorone®/ethinyl estradiol contraceptive vaginal ring (CVR); explore whether domains of use within the model predict satisfaction, method adherence and CVR continuation.
Study design: Four domains of use were considered relative to outcome markers of acceptability, that is, method satisfaction, adherence and continuation. A questionnaire to evaluate subjects' experiences relative to the domains, their satisfaction (Likert scale) and adherence to instructions for use was developed and administered to 1036 women enrolled in a 13-cycle Phase 3 trial. Method continuation was documented from the trial database. Stepwise logistic regression (LR) analysis was conducted and odds ratios (ORs) calculated to assess associations of satisfaction with questions from the four domains. Fisher's Exact Test was used to determine the association of satisfaction with outcome measures.
Results: A final acceptability model was developed based on the following determinants of CVR satisfaction: ease of use, side effects, expulsions/feeling the CVR and sexual activity including physical effects during intercourse. Satisfaction was high (89%) and related to higher method adherence [OR, 2.6 (1.3, 5.2)] and continuation [OR, 5.5 (3.5, 8.4)]. According to the LR analysis, attributes of CVR use representing items from the four domains - finding it easy to remove, not complaining of side effects, not feeling the CVR while wearing it and experiencing no change or an increase in sexual pleasure and/or frequency - were associated with higher odds of satisfaction.
Conclusion: Hypothesized domains of CVR use were related to satisfaction, which was associated with adherence and continuation. Results provide a scientific basis for introduction and future research.
Implications statement: Acceptability research is important when introducing a new method of contraception and determining whether it can be a successful option in meeting the reproductive health needs of women and men. This study was designed to test a conceptual model of acceptability and identify factors associated with successful use of a new contraceptive delivery modality. Original research was conducted for this publication.
Trial registration: ClinicalTrials.gov NCT00263341.
Keywords: Acceptability model; Hormonal contraception; Long-acting delivery system; Method introduction; Vaginal ring.
Copyright © 2014. Published by Elsevier Inc.
Figures
References
-
- Sivin I, Mishell DR, Alvarez F, et al. Contraceptive vaginal rings releasing Nestorone and ethinyl estradiol: a 1-year dose-finding trial. Contraception. 2005;71:122–9. - PubMed
-
- Brache V, Payán LJ, Faundes A. Current status of contraceptive vaginal rings. Contraception. 2013;87(3):264–72. - PubMed
-
- Faundes A, Hardy EE, Reyes Q, et al. Acceptability of the contraceptive vaginal ring by rural and urban populations in two Latin American countries. Contraception. 1981;24:393–414. - PubMed
-
- Darroch J, Sedgh G, Ball H. Contraceptives technologies: Responding to women’s needs. NewYork: Guttmacher Institute; 2011.
-
- Barot S. In search of breakthroughs: Renewing support for contraceptive research and development. Guttmacher Policy Review. 2013 Winter;16(1)
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical