Enhanced auto-antibody production and Mott cell formation in FcμR-deficient autoimmune mice
- PMID: 24994818
- PMCID: PMC4242241
- DOI: 10.1093/intimm/dxu070
Enhanced auto-antibody production and Mott cell formation in FcμR-deficient autoimmune mice
Abstract
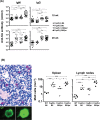
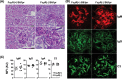
The IgM-Fc receptor (FcμR) is involved in IgM homeostasis as evidenced by increased pre-immune serum IgM and natural auto-antibodies of both IgM and IgG isotypes in Fcmr-deficient C57BL/6 (B6) mice. To determine the impact of Fcmr-ablation on autoimmunity, we introduced the Fcmr null mutation onto the Fas-deficient autoimmune-prone B6.MRL Fas (lpr/lpr) mouse background (B6/lpr). Both IgM and IgG auto-antibodies against dsDNA or chromatin appeared earlier in FcμR(-) B6/lpr than FcμR(+) B6/lpr mice, but this difference became less pronounced with age. Splenic B2 cells, which were 2-fold elevated in FcμR(+) B6/lpr mice, were reduced to normal B6 levels in FcμR(-) B6/lpr mice, whereas splenic B1 cells were comparable in both groups of B6/lpr mice. By contrast, marginal zone (MZ) B cells were markedly reduced in FcμR(-) B6/lpr mice compared with either FcμR(+) B6/lpr or wild type (WT) B6 mice. This reduction appeared to result from rapid differentiation of MZ B cells into plasma cells in the absence of FcμR, as IgM antibody to a Smith (Sm) antigen, to which MZ B cells are known to preferentially respond, was greatly increased in both groups (B6/lpr and B6) of FcμR(-) mice compared with FcμR(+) B6/lpr or B6 mice. Mott cells, aberrant plasma cells with intra-cytoplasmic inclusions, were also increased in the absence of FcμR. Despite these abnormalities, the severity of renal pathology and function and survival were all indistinguishable between FcμR(-) and FcμR(+) B6/lpr mice. Collectively, these findings suggest that FcμR plays important roles in the regulation of auto-antibody production, Mott cell formation and the differentiation of MZ B cells into plasma cells in B6.MRL Fas (lpr/lpr) mice.
Keywords: FcμR; IgM; Mott cells; auto-antibody; marginal zone B cells.
© The Japanese Society for Immunology. 2014. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures




Similar articles
-
Functional Roles of the IgM Fc Receptor in the Immune System.Front Immunol. 2019 May 3;10:945. doi: 10.3389/fimmu.2019.00945. eCollection 2019. Front Immunol. 2019. PMID: 31130948 Free PMC article. Review.
-
FcμR in human B cell subsets in primary selective IgM deficiency, and regulation of FcμR and production of natural IgM antibodies by IGIV.Hum Immunol. 2016 Dec;77(12):1194-1201. doi: 10.1016/j.humimm.2016.10.003. Epub 2016 Oct 14. Hum Immunol. 2016. PMID: 27751883 Free PMC article.
-
Altered Ig levels and antibody responses in mice deficient for the Fc receptor for IgM (FcμR).Proc Natl Acad Sci U S A. 2012 Sep 25;109(39):15882-7. doi: 10.1073/pnas.1206567109. Epub 2012 Sep 10. Proc Natl Acad Sci U S A. 2012. PMID: 22984178 Free PMC article.
-
Enhanced Mott cell formation linked with IgM Fc receptor (FcμR) deficiency.Eur J Immunol. 2023 Jul;53(7):e2250315. doi: 10.1002/eji.202250315. Epub 2023 May 11. Eur J Immunol. 2023. PMID: 37098762
-
Regulation of Humoral Immune Responses and B Cell Tolerance by the IgM Fc Receptor (FcμR).Adv Exp Med Biol. 2020;1254:75-86. doi: 10.1007/978-981-15-3532-1_7. Adv Exp Med Biol. 2020. PMID: 32323271 Review.
Cited by
-
Functional Roles of the IgM Fc Receptor in the Immune System.Front Immunol. 2019 May 3;10:945. doi: 10.3389/fimmu.2019.00945. eCollection 2019. Front Immunol. 2019. PMID: 31130948 Free PMC article. Review.
-
WT1 epitope-specific IgG and IgM antibodies for immune-monitoring in patients with advanced sarcoma treated with a WT1 peptide cancer vaccine.Oncol Lett. 2022 Feb;23(2):65. doi: 10.3892/ol.2022.13184. Epub 2022 Jan 3. Oncol Lett. 2022. PMID: 35069874 Free PMC article.
-
Distribution characteristics of FcμR positive cells in small intestinal lymph nodes of Bactrian camel.PLoS One. 2023 Jul 20;18(7):e0287329. doi: 10.1371/journal.pone.0287329. eCollection 2023. PLoS One. 2023. PMID: 37471384 Free PMC article.
-
Role of the IgM Fc Receptor in Immunity and Tolerance.Front Immunol. 2019 Mar 22;10:529. doi: 10.3389/fimmu.2019.00529. eCollection 2019. Front Immunol. 2019. PMID: 30967868 Free PMC article. Review.
-
Understanding Fc Receptor Involvement in Inflammatory Diseases: From Mechanisms to New Therapeutic Tools.Front Immunol. 2019 Apr 12;10:811. doi: 10.3389/fimmu.2019.00811. eCollection 2019. Front Immunol. 2019. PMID: 31057544 Free PMC article. Review.
References
-
- Ehrenstein M. R., Notley C. A. 2010. The importance of natural IgM: scavenger, protector and regulator. Nat. Rev. Immunol. 10:778. - PubMed
-
- Ravetch J. V., Nimmerjahn F. 2008. Fc receptors and their role in immune regulation and inflammation. In Paul W. E. ed., Fundamental Immunology, p. 684 Lippincott Williams & Wilkins, Philadelphia, PA, USA.
-
- Sanders S. K., Kubagawa H., Suzuki T., Butler J. L., Cooper M. D. 1987. IgM binding protein expressed by activated B cells. J. Immunol. 139:188. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

