Effects of location and timing of co-activated neurons in the auditory midbrain on cortical activity: implications for a new central auditory prosthesis
- PMID: 25003629
- PMCID: PMC7414896
- DOI: 10.1088/1741-2560/11/4/046021
Effects of location and timing of co-activated neurons in the auditory midbrain on cortical activity: implications for a new central auditory prosthesis
Abstract
Objective: An increasing number of deaf individuals are being implanted with central auditory prostheses, but their performance has generally been poorer than for cochlear implant users. The goal of this study is to investigate stimulation strategies for improving hearing performance with a new auditory midbrain implant (AMI). Previous studies have shown that repeated electrical stimulation of a single site in each isofrequency lamina of the central nucleus of the inferior colliculus (ICC) causes strong suppressive effects in elicited responses within the primary auditory cortex (A1). Here we investigate if improved cortical activity can be achieved by co-activating neurons with different timing and locations across an ICC lamina and if this cortical activity varies across A1.
Approach: We electrically stimulated two sites at different locations across an isofrequency ICC lamina using varying delays in ketamine-anesthetized guinea pigs. We recorded and analyzed spike activity and local field potentials across different layers and locations of A1.
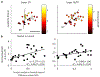
Results: Co-activating two sites within an isofrequency lamina with short inter-pulse intervals (<5 ms) could elicit cortical activity that is enhanced beyond a linear summation of activity elicited by the individual sites. A significantly greater extent of normalized cortical activity was observed for stimulation of the rostral-lateral region of an ICC lamina compared to the caudal-medial region. We did not identify any location trends across A1, but the most cortical enhancement was observed in supragranular layers, suggesting further integration of the stimuli through the cortical layers.
Significance: The topographic organization identified by this study provides further evidence for the presence of functional zones across an ICC lamina with locations consistent with those identified by previous studies. Clinically, these results suggest that co-activating different neural populations in the rostral-lateral ICC rather than the caudal-medial ICC using the AMI may improve or elicit different types of hearing capabilities.
Conflict of interest statement
Conflicts of interest
None.
Figures
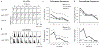
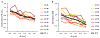
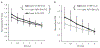
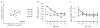
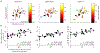


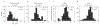
Similar articles
-
Auditory midbrain implant: research and development towards a second clinical trial.Hear Res. 2015 Apr;322:212-23. doi: 10.1016/j.heares.2015.01.006. Epub 2015 Jan 20. Hear Res. 2015. PMID: 25613994 Free PMC article. Review.
-
Neural integration and enhancement from the inferior colliculus up to different layers of auditory cortex.J Neurophysiol. 2013 Aug;110(4):1009-20. doi: 10.1152/jn.00022.2013. Epub 2013 May 29. J Neurophysiol. 2013. PMID: 23719210 Free PMC article.
-
Spatially distinct functional output regions within the central nucleus of the inferior colliculus: implications for an auditory midbrain implant.J Neurosci. 2007 Aug 8;27(32):8733-43. doi: 10.1523/JNEUROSCI.5127-06.2007. J Neurosci. 2007. PMID: 17687050 Free PMC article.
-
Coactivation of different neurons within an isofrequency lamina of the inferior colliculus elicits enhanced auditory cortical activation.J Neurophysiol. 2012 Aug;108(4):1199-210. doi: 10.1152/jn.00111.2012. Epub 2012 May 23. J Neurophysiol. 2012. PMID: 22623485
-
Brief and short-term corticofugal modulation of acoustic signal processing in the bat midbrain.Hear Res. 2002 Jun;168(1-2):196-207. doi: 10.1016/s0378-5955(02)00358-1. Hear Res. 2002. PMID: 12117521 Review.
Cited by
-
Auditory midbrain implant: research and development towards a second clinical trial.Hear Res. 2015 Apr;322:212-23. doi: 10.1016/j.heares.2015.01.006. Epub 2015 Jan 20. Hear Res. 2015. PMID: 25613994 Free PMC article. Review.
-
Suppression and facilitation of auditory neurons through coordinated acoustic and midbrain stimulation: investigating a deep brain stimulator for tinnitus.J Neural Eng. 2014 Dec;11(6):066001. doi: 10.1088/1741-2560/11/6/066001. Epub 2014 Oct 13. J Neural Eng. 2014. PMID: 25307351 Free PMC article.
-
Hearing the light: neural and perceptual encoding of optogenetic stimulation in the central auditory pathway.Sci Rep. 2015 May 22;5:10319. doi: 10.1038/srep10319. Sci Rep. 2015. PMID: 26000557 Free PMC article.
References
-
- Shannon RV, Zeng FG, Kamath V, Wygonski J and Ekelid M 1995. Speech recognition with primarily temporal cues Science 270 303–4 - PubMed
-
- Kral A and O’Donoghue GM 2010. Profound deafness in childhood N. Engl. J. Med 3 1438–50 - PubMed
-
- Colletti V, Shannon RV, Carner M, Veronese S and Colletti L 2009. Progress in restoration of hearing with the auditory brainstem implant Prog. Brain Res 175 333–45 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
