Structural basis for cooperative interactions of substituted 2-aminopyrimidines with the acetylcholine binding protein
- PMID: 25006260
- PMCID: PMC4115551
- DOI: 10.1073/pnas.1410992111
Structural basis for cooperative interactions of substituted 2-aminopyrimidines with the acetylcholine binding protein
Abstract
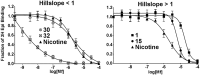
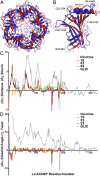
The nicotinic acetylcholine receptor (nAChR) and the acetylcholine binding protein (AChBP) are pentameric oligomers in which binding sites for nicotinic agonists and competitive antagonists are found at selected subunit interfaces. The nAChR spontaneously exists in multiple conformations associated with its activation and desensitization steps, and conformations are selectively stabilized by binding of agonists and antagonists. In the nAChR, agonist binding and the associated conformational changes accompanying activation and desensitization are cooperative. AChBP, which lacks the transmembrane spanning and cytoplasmic domains, serves as a homology model of the extracellular domain of the nAChRs. We identified unique cooperative binding behavior of a number of 4,6-disubstituted 2-aminopyrimidines to Lymnaea AChBP, with different molecular variants exhibiting positive, nH > 1.0, and negative cooperativity, nH < 1.0. Therefore, for a distinctive set of ligands, the extracellular domain of a nAChR surrogate suffices to accommodate cooperative interactions. X-ray crystal structures of AChBP complexes with examples of each allowed the identification of structural features in the ligands that confer differences in cooperative behavior. Both sets of molecules bind at the agonist-antagonist site, as expected from their competition with epibatidine. An analysis of AChBP quaternary structure shows that cooperative ligand binding is associated with a blooming or flare conformation, a structural change not observed with the classical, noncooperative, nicotinic ligands. Positively and negatively cooperative ligands exhibited unique features in the detailed binding determinants and poses of the complexes.
Keywords: allostery; crystallography; nicotinic receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

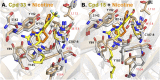
Similar articles
-
Substituted 2-Aminopyrimidines Selective for α7-Nicotinic Acetylcholine Receptor Activation and Association with Acetylcholine Binding Proteins.J Am Chem Soc. 2017 Mar 15;139(10):3676-3684. doi: 10.1021/jacs.6b10746. Epub 2017 Mar 1. J Am Chem Soc. 2017. PMID: 28221788
-
Galanthamine and non-competitive inhibitor binding to ACh-binding protein: evidence for a binding site on non-alpha-subunit interfaces of heteromeric neuronal nicotinic receptors.J Mol Biol. 2007 Jun 15;369(4):895-901. doi: 10.1016/j.jmb.2007.03.067. Epub 2007 Mar 31. J Mol Biol. 2007. PMID: 17481657 Free PMC article.
-
Complex between α-bungarotoxin and an α7 nicotinic receptor ligand-binding domain chimaera.Biochem J. 2013 Sep 1;454(2):303-310. doi: 10.1042/BJ20130636. Biochem J. 2013. PMID: 23800261 Free PMC article.
-
Structure and function of AChBP, homologue of the ligand-binding domain of the nicotinic acetylcholine receptor.Ann N Y Acad Sci. 2003 Sep;998:81-92. doi: 10.1196/annals.1254.010. Ann N Y Acad Sci. 2003. PMID: 14592865 Review.
-
The 2.7 A structure of AChBP, homologue of the ligand-binding domain of the nicotinic acetylcholine receptor.Novartis Found Symp. 2002;245:22-9; discussion 29-32, 165-8. Novartis Found Symp. 2002. PMID: 12027010 Review.
Cited by
-
Modulation of cholinergic activity through lynx prototoxins: Implications for cognition and anxiety regulation.Neuropharmacology. 2020 Sep 1;174:108071. doi: 10.1016/j.neuropharm.2020.108071. Epub 2020 Apr 13. Neuropharmacology. 2020. PMID: 32298703 Free PMC article. Review. No abstract available.
-
Cooperative binding: a multiple personality.J Math Biol. 2016 Jun;72(7):1747-74. doi: 10.1007/s00285-015-0922-z. Epub 2015 Aug 29. J Math Biol. 2016. PMID: 26319983
-
Probing function in ligand-gated ion channels without measuring ion transport.J Gen Physiol. 2022 Jun 6;154(6):e202213082. doi: 10.1085/jgp.202213082. Epub 2022 May 25. J Gen Physiol. 2022. PMID: 35612603 Free PMC article.
-
Mechanism of calcium potentiation of the α7 nicotinic acetylcholine receptor.J Gen Physiol. 2020 Sep 7;152(9):e202012606. doi: 10.1085/jgp.202012606. J Gen Physiol. 2020. PMID: 32702089 Free PMC article.
-
Full and partial agonists evoke distinct structural changes in opening the muscle acetylcholine receptor channel.J Gen Physiol. 2018 May 7;150(5):713-729. doi: 10.1085/jgp.201711881. Epub 2018 Apr 21. J Gen Physiol. 2018. PMID: 29680816 Free PMC article.
References
-
- Changeux JP. 50 years of allosteric interactions: The twists and turns of the models. Nat Rev Mol Cell Biol. 2013;14(12):819–829. - PubMed
-
- Karlin A. Emerging structure of the nicotinic acetylcholine receptors. Nat Rev Neurosci. 2002;3(2):102–114. - PubMed
-
- Thompson AJ, Lester HA, Lummis SC. The structural basis of function in Cys-loop receptors. Q Rev Biophys. 2010;43(4):449–499. - PubMed
-
- Corringer PJ, et al. Structure and pharmacology of pentameric receptor channels: From bacteria to brain. Structure. 2012;20(6):941–956. - PubMed
-
- Heidmann T, Changeux JP. Fast kinetic studies on the interaction of a fluorescent agonist with the membrane-bound acetylcholine receptor from Torpedo marmorata. Eur J Biochem. 1979;94(1):255–279. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

