Dual modulation of type I interferon response by bluetongue virus
- PMID: 25008919
- PMCID: PMC4178850
- DOI: 10.1128/JVI.01235-14
Dual modulation of type I interferon response by bluetongue virus
Abstract
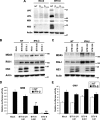
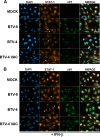
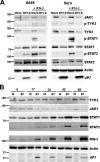
Bluetongue virus (BTV) is a double-stranded RNA (dsRNA) virus that causes an economically important disease in ruminants. BTV infection is a strong inducer of type I interferon (IFN-I) in multiple cell types. It has been shown recently that BTV and, more specifically, the nonstructural protein NS3 of BTV are able to modulate the IFN-I synthesis pathway. However, nothing is known about the ability of BTV to counteract IFN-I signaling. Here, we investigated the effect of BTV on the IFN-I response pathway and, more particularly, the Janus tyrosine kinase (JAK)/signal transducer and activator of transcription protein (STAT) signaling pathway. We found that BTV infection triggered the expression of IFN-stimulated genes (ISGs) in A549 cells. However, when BTV-infected cells were stimulated with external IFN-I, we showed that activation of the IFN-stimulated response element (ISRE) promoter and expression of ISGs were inhibited. We found that this inhibition involved two different mechanisms that were dependent on the time of infection. After overnight infection, BTV blocked specifically the phosphorylation and nuclear translocation of STAT1. This inhibition correlated with the redistribution of STAT1 in regions adjacent to the nucleus. At a later time point of infection, BTV was found to interfere with the activation of other key components of the JAK/STAT pathway and to induce the downregulation of JAK1 and TYK2 protein expression. Overall, our study indicates for the first time that BTV is able to interfere with the JAK/STAT pathway to modulate the IFN-I response.
Importance: Bluetongue virus (BTV) causes a severe disease in ruminants and has an important impact on the livestock economy in areas of endemicity such as Africa. The emergence of strains, such as serotype 8 in Europe in 2006, can lead to important economic losses due to commercial restrictions and prophylactic measures. It has been known for many years that BTV is a strong inducer of type I interferon (IFN-I) in vitro and in vivo in multiple cell types. However, the ability of BTV to interact with the IFN-I system remains unclear. Here, we report that BTV is able to modulate the IFN-I response by interfering with the Janus tyrosine kinase (JAK)/signal transducer and activator of transcription protein (STAT) signaling pathway. These findings contribute to knowledge of how BTV infection interferes with the host's innate immune response and becomes pathogenic. This will also be important for the design of efficacious vaccine candidates.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures




Similar articles
-
Bluetongue Virus NS4 Protein Is an Interferon Antagonist and a Determinant of Virus Virulence.J Virol. 2016 May 12;90(11):5427-39. doi: 10.1128/JVI.00422-16. Print 2016 Jun 1. J Virol. 2016. PMID: 27009961 Free PMC article.
-
Bluetongue virus non-structural protein 3 (NS3) and NS4 coordinatively antagonize type Ⅰ interferon signaling by targeting STAT1.Vet Microbiol. 2021 Mar;254:108986. doi: 10.1016/j.vetmic.2021.108986. Epub 2021 Jan 12. Vet Microbiol. 2021. PMID: 33486325
-
Novel Function of Bluetongue Virus NS3 Protein in Regulation of the MAPK/ERK Signaling Pathway.J Virol. 2019 Jul 30;93(16):e00336-19. doi: 10.1128/JVI.00336-19. Print 2019 Aug 15. J Virol. 2019. PMID: 31167915 Free PMC article.
-
Induction and control of the type I interferon pathway by Bluetongue virus.Virus Res. 2014 Mar;182:59-70. doi: 10.1016/j.virusres.2013.10.027. Epub 2013 Nov 7. Virus Res. 2014. PMID: 24211608 Free PMC article. Review.
-
Interferon α/β receptor knockout mice as a model to study bluetongue virus infection.Virus Res. 2014 Mar;182:35-42. doi: 10.1016/j.virusres.2013.09.038. Epub 2013 Oct 4. Virus Res. 2014. PMID: 24100234 Review.
Cited by
-
Virus-induced autophagic degradation of STAT2 as a mechanism for interferon signaling blockade.EMBO Rep. 2019 Nov 5;20(11):e48766. doi: 10.15252/embr.201948766. Epub 2019 Oct 11. EMBO Rep. 2019. PMID: 31603272 Free PMC article.
-
Characterization of a second open reading frame in genome segment 10 of bluetongue virus.J Gen Virol. 2015 Nov;96(11):3280-3293. doi: 10.1099/jgv.0.000267. Epub 2015 Aug 19. J Gen Virol. 2015. PMID: 26290332 Free PMC article.
-
The Amino-Terminal Region of Hepatitis E Virus ORF1 Containing a Methyltransferase (Met) and a Papain-Like Cysteine Protease (PCP) Domain Counteracts Type I Interferon Response.Viruses. 2018 Dec 18;10(12):726. doi: 10.3390/v10120726. Viruses. 2018. PMID: 30567349 Free PMC article.
-
Double-stranded RNA orbivirus disrupts the DNA-sensing cGAS-sting axis to prevent type I IFN induction.Cell Mol Life Sci. 2025 Jan 21;82(1):55. doi: 10.1007/s00018-025-05580-5. Cell Mol Life Sci. 2025. PMID: 39836220 Free PMC article.
-
Bluetongue Virus NS4 Protein Is an Interferon Antagonist and a Determinant of Virus Virulence.J Virol. 2016 May 12;90(11):5427-39. doi: 10.1128/JVI.00422-16. Print 2016 Jun 1. J Virol. 2016. PMID: 27009961 Free PMC article.
References
-
- Mertens PPC, Diprose J, Maan S, Singh KP, Attoui H, Samuel AR. 2004. Bluetongue virus replication, molecular and structural biology. Vet. Ital. 40:426–437 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

