Virus-like particle secretion and genotype-dependent immunogenicity of dengue virus serotype 2 DNA vaccine
- PMID: 25008922
- PMCID: PMC4178884
- DOI: 10.1128/JVI.00810-14
Virus-like particle secretion and genotype-dependent immunogenicity of dengue virus serotype 2 DNA vaccine
Abstract
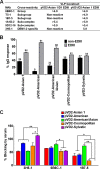
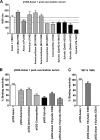
Dengue virus (DENV), composed of four distinct serotypes, is the most important and rapidly emerging arthropod-borne pathogen and imposes substantial economic and public health burdens. We constructed candidate vaccines containing the DNA of five of the genotypes of dengue virus serotype 2 (DENV-2) and evaluated the immunogenicity, the neutralizing (Nt) activity of the elicited antibodies, and the protective efficacy elicited in mice immunized with the vaccine candidates. We observed a significant correlation between the level of in vitro virus-like particle secretion, the elicited antibody response, and the protective efficacy of the vaccines containing the DNA of the different DENV genotypes in immunized mice. However, higher total IgG antibody levels did not always translate into higher Nt antibodies against homologous and heterologous viruses. We also found that, in contrast to previous reports, more than 50% of total IgG targeted ectodomain III (EDIII) of the E protein, and a substantial fraction of this population was interdomain highly neutralizing flavivirus subgroup-cross-reactive antibodies, such as monoclonal antibody 1B7-5. In addition, the lack of a critical epitope(s) in the Sylvatic genotype virus recognized by interdomain antibodies could be the major cause of the poor protection of mice vaccinated with the Asian 1 genotype vaccine (pVD2-Asian 1) from lethal challenge with virus of the Sylvatic genotype. In conclusion, although the pVD2-Asian 1 vaccine was immunogenic, elicited sufficient titers of Nt antibodies against all DENV-2 genotypes, and provided 100% protection against challenge with virus of the homologous Asian 1 genotype and virus of the heterologous Cosmopolitan genotype, it is critical to monitor the potential emergence of Sylvatic genotype viruses, since vaccine candidates under development may not protect vaccinated humans from these viruses.
Importance: Five genotype-specific dengue virus serotype 2 (DENV-2) DNA vaccine candidates were evaluated for their immunogenicity, homologous and heterologous neutralizing (Nt) antibody titers, and cross-genotype protection in a murine model. The immunity elicited by our prototype vaccine candidate (Asian 1 genotype strain 16681) in mice was protective against viruses of other genotypes but not against virus of the Sylvatic genotype, whose emergence and potential risk after introduction into the human population have previously been demonstrated. The underlying mechanism of a lack of protection elicited by the prototype vaccine may at least be contributed by the absence of a flavivirus subgroup-cross-reactive, highly neutralizing monoclonal antibody 1B7-5-like epitope in DENV-2 of the Sylvatic genotype. The DENV DNA vaccine directs the synthesis and assembly of virus-like particles (VLPs) and induces immune responses similar to those elicited by live-attenuated vaccines, and its flexibility permits the fast deployment of vaccine to combat emerging viruses, such as Sylvatic genotype viruses. The enhanced VLP secretion obtained by replacement of ectodomain I-II (EDI-II) of the Cosmopolitan genotype vaccine construct (VD2-Cosmopolitan) with the Asian 1 EDI-II elicited significantly higher total IgG and Nt antibody titers and suggests a novel approach to enhance the immunogenicity of the DNA vaccine. A DENV vaccine capable of eliciting protective immunity against viruses of existing and emerging genotypes should be the focus of future DENV vaccine development.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures




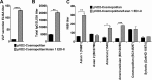
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

