Dissecting the signaling mechanisms underlying recognition and preference of food odors
- PMID: 25009271
- PMCID: PMC4087214
- DOI: 10.1523/JNEUROSCI.0012-14.2014
Dissecting the signaling mechanisms underlying recognition and preference of food odors
Abstract
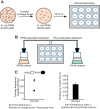
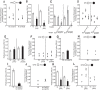
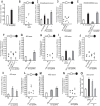
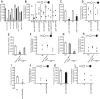
Food is critical for survival. Many animals, including the nematode Caenorhabditis elegans, use sensorimotor systems to detect and locate preferred food sources. However, the signaling mechanisms underlying food-choice behaviors are poorly understood. Here, we characterize the molecular signaling that regulates recognition and preference between different food odors in C. elegans. We show that the major olfactory sensory neurons, AWB and AWC, play essential roles in this behavior. A canonical Gα-protein, together with guanylate cyclases and cGMP-gated channels, is needed for the recognition of food odors. The food-odor-evoked signal is transmitted via glutamatergic neurotransmission from AWC and through AMPA and kainate-like glutamate receptor subunits. In contrast, peptidergic signaling is required to generate preference between different food odors while being dispensable for the recognition of the odors. We show that this regulation is achieved by the neuropeptide NLP-9 produced in AWB, which acts with its putative receptor NPR-18, and by the neuropeptide NLP-1 produced in AWC. In addition, another set of sensory neurons inhibits food-odor preference. These mechanistic logics, together with a previously mapped neural circuit underlying food-odor preference, provide a functional network linking sensory response, transduction, and downstream receptors to process complex olfactory information and generate the appropriate behavioral decision essential for survival.
Keywords: glutamatergic transmission; neuropeptide signaling; olfactory sensory neurons; olfactory sensory signaling; preference of food odors.
Copyright © 2014 the authors 0270-6474/14/339389-15$15.00/0.
Figures



References
-
- Berkowicz DA, Trombley PQ, Shepherd GM. Evidence for glutamate as the olfactory receptor cell neurotransmitter. J Neurophysiol. 1994;71:2557–2561. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
