Differential GABAergic and glycinergic inputs of inhibitory interneurons and Purkinje cells to principal cells of the cerebellar nuclei
- PMID: 25009273
- PMCID: PMC6608357
- DOI: 10.1523/JNEUROSCI.0401-14.2014
Differential GABAergic and glycinergic inputs of inhibitory interneurons and Purkinje cells to principal cells of the cerebellar nuclei
Abstract
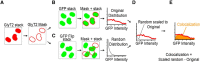
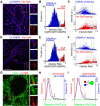
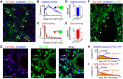
The principal neurons of the cerebellar nuclei (CN), the sole output of the olivo-cerebellar system, receive a massive inhibitory input from Purkinje cells (PCs) of the cerebellar cortex. Morphological evidence suggests that CN principal cells are also contacted by inhibitory interneurons, but the properties of this connection are unknown. Using transgenic, tracing, and immunohistochemical approaches in mice, we show that CN interneurons form a large heterogeneous population with GABA/glycinergic phenotypes, distinct from GABAergic olive-projecting neurons. CN interneurons are found to contact principal output neurons, via glycine receptor (GlyR)-enriched synapses, virtually devoid of the main GABA receptor (GABAR) subunits α1 and γ2. Those clusters account for 5% of the total number of inhibitory receptor clusters on principal neurons. Brief optogenetic stimulations of CN interneurons, through selective expression of channelrhodopsin 2 after viral-mediated transfection of the flexed gene in GlyT2-Cre transgenic mice, evoked fast IPSCs in principal cells. GlyR activation accounted for 15% of interneuron IPSC amplitude, while the remaining current was mediated by activation of GABAR. Surprisingly, small GlyR clusters were also found at PC synapses onto principal CN neurons in addition to α1 and γ2 GABAR subunits. However, GlyR activation was found to account for <3% of the PC inhibitory synaptic currents evoked by electrical stimulation. This work establishes CN glycinergic neurons as a significant source of inhibition to CN principal cells, forming contacts molecularly distinct from, but functionally similar to, Purkinje cell synapses. Their impact on CN output, motor learning, and motor execution deserves further investigation.
Keywords: cerebellar nuclei; glycinergic; immunohistochemistry; interneurons; mixed inhibition; optogenetics.
Copyright © 2014 the authors 0270-6474/14/349418-14$15.00/0.
Figures




References
-
- Bäurle J, Grüsser-Cornehls U. Differential number of glycine- and GABA-immunopositive neurons and terminals in the deep cerebellar nuclei of normal and Purkinje cell degeneration mutant mice. J Comp Neurol. 1997;382:443–458. doi: 10.1002/(SICI)1096-9861(19970616)382:4<443::AID-CNE2>3.0.CO;2-2. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
