Synthetic Routes to Methylerythritol Phosphate Pathway Intermediates and Downstream Isoprenoids
- PMID: 25009443
- PMCID: PMC4082188
- DOI: 10.2174/1385272819666140501001101
Synthetic Routes to Methylerythritol Phosphate Pathway Intermediates and Downstream Isoprenoids
Abstract
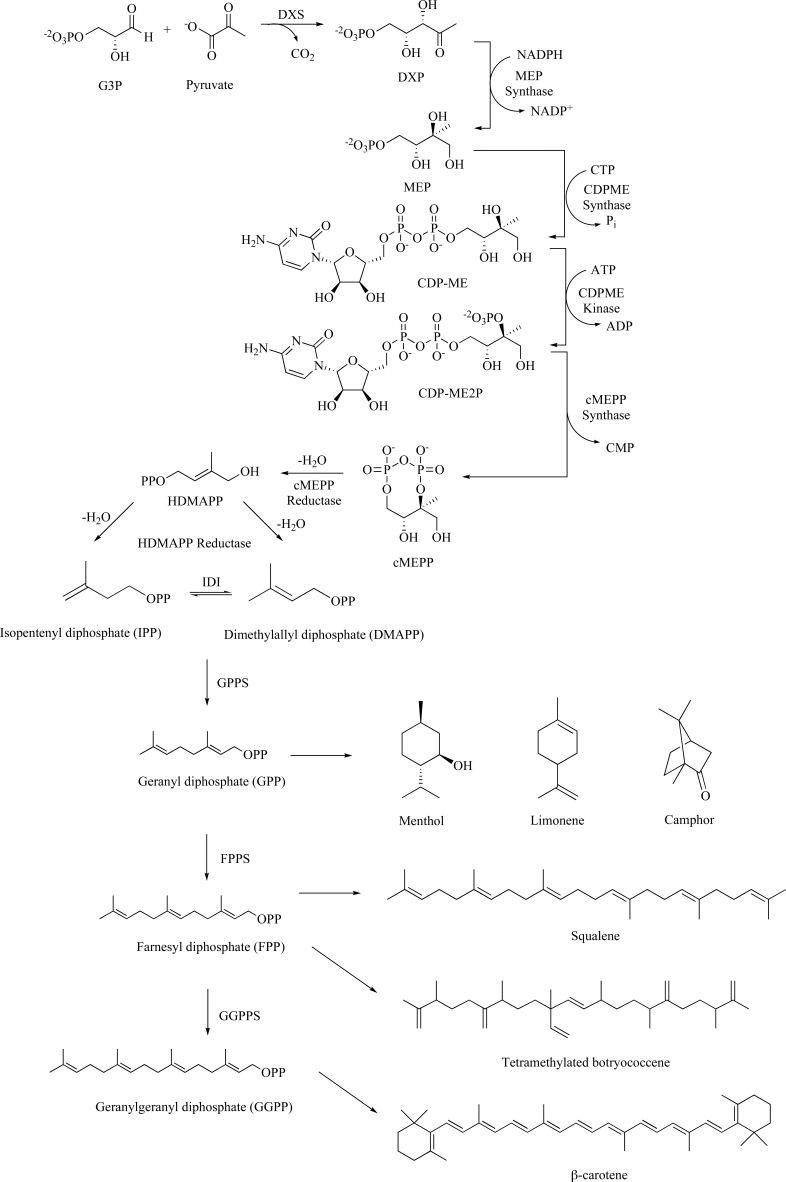
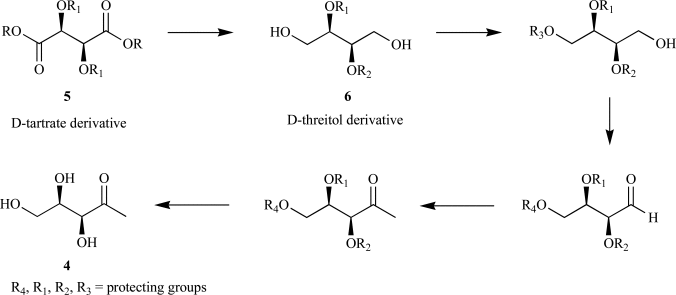
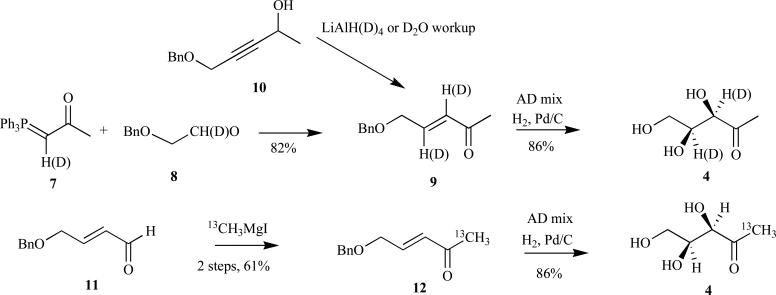
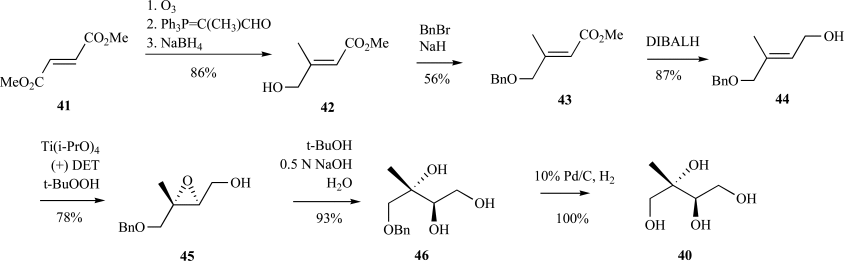
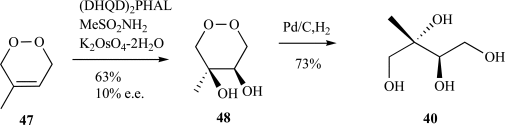
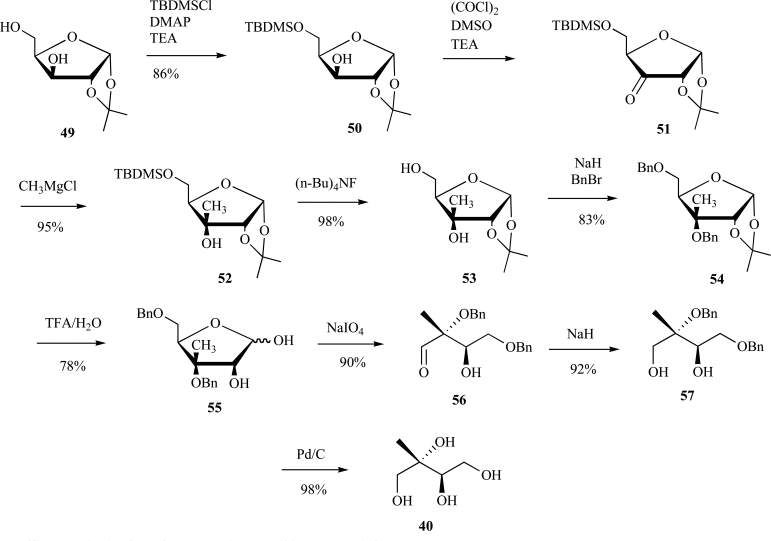
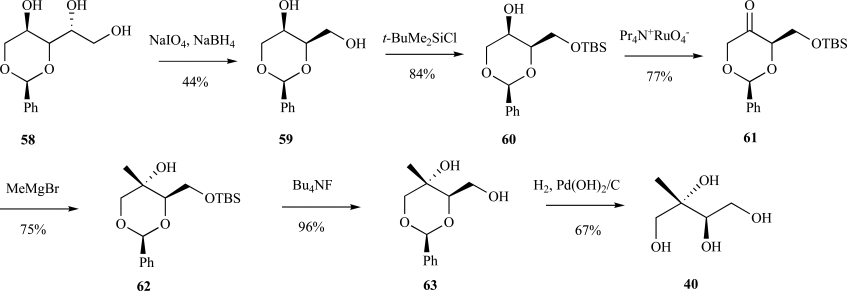
Isoprenoids constitute the largest class of natural products with greater than 55,000 identified members. They play essential roles in maintaining proper cellular function leading to maintenance of human health, plant defense mechanisms against predators, and are often exploited for their beneficial properties in the pharmaceutical and nutraceutical industries. Most impressively, all known isoprenoids are derived from one of two C5-precursors, isopentenyl diphosphate (IPP) or dimethylallyl diphosphate (DMAPP). In order to study the enzyme transformations leading to the extensive structural diversity found within this class of compounds there must be access to the substrates. Sometimes, intermediates within a biological pathway can be isolated and used directly to study enzyme/pathway function. However, the primary route to most of the isoprenoid intermediates is through chemical catalysis. As such, this review provides the first exhaustive examination of synthetic routes to isoprenoid and isoprenoid precursors with particular emphasis on the syntheses of intermediates found as part of the 2C-methylerythritol 4-phosphate (MEP) pathway. In addition, representative syntheses are presented for the monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), triterpenes (C30) and tetraterpenes (C40). Finally, in some instances, the synthetic routes to substrate analogs found both within the MEP pathway and downstream isoprenoids are examined.
Keywords: Enzyme mechanism; MEP pathway.; isoprenoids; terpenes.
Figures












Similar articles
-
Both methylerythritol phosphate and mevalonate pathways contribute to biosynthesis of each of the major isoprenoid classes in young cotton seedlings.Phytochemistry. 2014 Feb;98:110-9. doi: 10.1016/j.phytochem.2013.11.010. Epub 2013 Dec 17. Phytochemistry. 2014. PMID: 24359633
-
Methylerythritol phosphate pathway of isoprenoid biosynthesis.Annu Rev Biochem. 2013;82:497-530. doi: 10.1146/annurev-biochem-052010-100934. Annu Rev Biochem. 2013. PMID: 23746261 Free PMC article. Review.
-
Terpenes and isoprenoids: a wealth of compounds for global use.Planta. 2019 Jan;249(1):1-8. doi: 10.1007/s00425-018-3056-x. Epub 2018 Nov 22. Planta. 2019. PMID: 30467631 Review.
-
Current development in isoprenoid precursor biosynthesis and regulation.Curr Opin Chem Biol. 2013 Aug;17(4):571-9. doi: 10.1016/j.cbpa.2013.06.020. Epub 2013 Jul 24. Curr Opin Chem Biol. 2013. PMID: 23891475 Free PMC article. Review.
-
MEP pathway products allosterically promote monomerization of deoxy-D-xylulose-5-phosphate synthase to feedback-regulate their supply.Plant Commun. 2023 May 8;4(3):100512. doi: 10.1016/j.xplc.2022.100512. Epub 2022 Dec 26. Plant Commun. 2023. PMID: 36575800 Free PMC article.
Cited by
-
Regulatory microRNAs and phasiRNAs of paclitaxel biosynthesis in Taxus chinensis.Front Plant Sci. 2024 May 8;15:1403060. doi: 10.3389/fpls.2024.1403060. eCollection 2024. Front Plant Sci. 2024. PMID: 38779066 Free PMC article.
-
Recent Research Progress in Taxol Biosynthetic Pathway and Acylation Reactions Mediated by Taxus Acyltransferases.Molecules. 2021 May 12;26(10):2855. doi: 10.3390/molecules26102855. Molecules. 2021. PMID: 34065782 Free PMC article. Review.
-
Genetic resources for advanced biofuel production described with the Gene Ontology.Front Microbiol. 2014 Oct 10;5:528. doi: 10.3389/fmicb.2014.00528. eCollection 2014. Front Microbiol. 2014. PMID: 25346727 Free PMC article. Review.
References
-
- Bach TJ, Rohmer M. Isoprenoid Synthesis in Plants and Microorganisms. Springer: New York. 2012:505.
-
- Chappell J. Biochemistry and molecular biology of the isoprenoid biosynthetic pathway in plants. Annu. Rev. Plant. Phys. 1995;46:521–547.
-
- Kirby J, Keasling JD. Biosynthesis of plant isoprenoids: Perspectives for microbial engineering. Annu. Rev. Plant. Biol. 2009;60:335–355. - PubMed
-
- Eubanks LM, Poulter CD. Rhodobacter capsulatus DXP synthase: Steady-state kinetics and substrate binding. Biochemistry. 2001;40:8613–8614. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
