Artificial symbiosis for acetone-butanol-ethanol (ABE) fermentation from alkali extracted deshelled corn cobs by co-culture of Clostridium beijerinckii and Clostridium cellulovorans
- PMID: 25023325
- PMCID: PMC4223388
- DOI: 10.1186/s12934-014-0092-5
Artificial symbiosis for acetone-butanol-ethanol (ABE) fermentation from alkali extracted deshelled corn cobs by co-culture of Clostridium beijerinckii and Clostridium cellulovorans
Abstract
Background: Butanol is an industrial commodity and also considered to be a more promising gasoline substitute compared to ethanol. Renewed attention has been paid to solvents (acetone, butanol and ethanol) production from the renewable and inexpensive substrates, for example, lignocellulose, on account of the depletion of oil resources, increasing gasoline prices and deteriorating environment. Limited to current tools for genetic manipulation, it is difficult to develop a genetically engineered microorganism with combined ability of lignocellulose utilization and solvents production. Mixed culture of cellulolytic microorganisms and solventogenic bacteria provides a more convenient and feasible approach for ABE fermentation due to the potential for synergistic utilization of the metabolic pathways of two organisms. But few bacteria pairs succeeded in producing biobutanol of high titer or high productivity without adding butyrate. The aim of this work was to use Clostridium cellulovorans 743B to saccharify lignocellulose and produce butyric acid, instead of adding cellulase and butyric acid to the medium, so that the soluble sugars and butyric acid generated can be subsequently utilized by Clostridium beijerinckii NCIMB 8052 to produce butanol in one pot reaction.
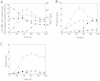
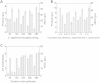
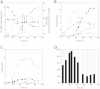
Results: A stable artificial symbiotic system was constructed by co-culturing a celluloytic, anaerobic, butyrate-producing mesophile (C. cellulovorans 743B) and a non-celluloytic, solventogenic bacterium (C. beijerinckii NCIMB 8052) to produce solvents by consolidated bioprocessing (CBP) with alkali extracted deshelled corn cobs (AECC), a low-cost renewable feedstock, as the sole carbon source. Under optimized conditions, the co-culture degraded 68.6 g/L AECC and produced 11.8 g/L solvents (2.64 g/L acetone, 8.30 g/L butanol and 0.87 g/L ethanol) in less than 80 h. Besides, a real-time PCR assay based on the 16S rRNA gene sequence was performed to study the dynamics of the abundance of each strain during the co-culturing process, which figured out the roles of each strain at different periods in the symbiosis.
Conclusion: Our work illustrated the great potential of artificial symbiosis in biofuel production from lignocellulosic biomass by CBP. The dynamics of the abundance of C. beijerinckii and C. cellulovorans revealed mechanisms of cooperation and competition between the two strains during the co-culture process.
Figures

Similar articles
-
Improved n-Butanol Production from Clostridium cellulovorans by Integrated Metabolic and Evolutionary Engineering.Appl Environ Microbiol. 2019 Mar 22;85(7):e02560-18. doi: 10.1128/AEM.02560-18. Print 2019 Apr 1. Appl Environ Microbiol. 2019. PMID: 30658972 Free PMC article.
-
Enhanced solvent production by metabolic engineering of a twin-clostridial consortium.Metab Eng. 2017 Jan;39:38-48. doi: 10.1016/j.ymben.2016.10.013. Epub 2016 Oct 27. Metab Eng. 2017. PMID: 27794465
-
Utilization of banana crop residue as an agricultural bioresource for the production of acetone-butanol-ethanol by Clostridium beijerinckii YVU1.Lett Appl Microbiol. 2020 Jan;70(1):36-41. doi: 10.1111/lam.13239. Epub 2019 Dec 3. Lett Appl Microbiol. 2020. PMID: 31631376
-
Butanol fermentation research: upstream and downstream manipulations.Chem Rec. 2004;4(5):305-14. doi: 10.1002/tcr.20023. Chem Rec. 2004. PMID: 15543610 Review.
-
Smart fermentation engineering for butanol production: designed biomass and consolidated bioprocessing systems.Appl Microbiol Biotechnol. 2019 Dec;103(23-24):9359-9371. doi: 10.1007/s00253-019-10198-2. Epub 2019 Nov 13. Appl Microbiol Biotechnol. 2019. PMID: 31720773 Review.
Cited by
-
Design and validation of a multiplex PCR method for the simultaneous quantification of Clostridium acetobutylicum, Clostridium carboxidivorans and Clostridium cellulovorans.Sci Rep. 2023 Nov 16;13(1):20073. doi: 10.1038/s41598-023-47007-w. Sci Rep. 2023. PMID: 37973932 Free PMC article.
-
Comprehensive evaluation for the one-pot biosynthesis of butyl acetate by using microbial mono- and co-cultures.Biotechnol Biofuels. 2021 Oct 16;14(1):203. doi: 10.1186/s13068-021-02053-2. Biotechnol Biofuels. 2021. PMID: 34656153 Free PMC article.
-
Flow cytometry analysis of Clostridium beijerinckii NRRL B-598 populations exhibiting different phenotypes induced by changes in cultivation conditions.Biotechnol Biofuels. 2018 Apr 6;11:99. doi: 10.1186/s13068-018-1096-x. eCollection 2018. Biotechnol Biofuels. 2018. PMID: 29632557 Free PMC article.
-
Current progress on engineering microbial strains and consortia for production of cellulosic butanol through consolidated bioprocessing.Microb Biotechnol. 2023 Feb;16(2):238-261. doi: 10.1111/1751-7915.14148. Epub 2022 Sep 27. Microb Biotechnol. 2023. PMID: 36168663 Free PMC article. Review.
-
Consolidated bioprocessing for butanol production of cellulolytic Clostridia: development and optimization.Microb Biotechnol. 2020 Mar;13(2):410-422. doi: 10.1111/1751-7915.13478. Epub 2019 Aug 26. Microb Biotechnol. 2020. PMID: 31448546 Free PMC article. Review.
References
-
- Qureshi N, Saha B, Cotta M: Butanol production from wheat straw hydrolysate usingClostridium beijerinckii.Bioprocess Biosyst Eng 2007, 30:419–427. - PubMed
-
- Qureshi N, Ezeji T, Ebener J, Dien B, Cotta M, Blaschek H: Butanol production byClostridium beijerinckii. Part I: Use of acid and enzyme hydrolyzed corn fiber.Bioresour Technol 2008, 99:5915–5922. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

