The JDRF CCTN CGM TIME Trial: Timing of Initiation of continuous glucose Monitoring in Established pediatric type 1 diabetes: study protocol, recruitment and baseline characteristics
- PMID: 25034216
- PMCID: PMC4109785
- DOI: 10.1186/1471-2431-14-183
The JDRF CCTN CGM TIME Trial: Timing of Initiation of continuous glucose Monitoring in Established pediatric type 1 diabetes: study protocol, recruitment and baseline characteristics
Abstract
Background: Continuous glucose monitoring (CGM) has been shown to improve glucose control in adults with type 1 diabetes. Effectiveness of CGM is directly linked with CGM adherence, which can be challenging to maintain in children and adolescents. We hypothesize that initiating CGM at the same time as starting insulin pump therapy in pump naïve children and adolescents with type 1 diabetes will result in greater CGM adherence and effectiveness compared to delaying CGM introduction by 6 months, and that this is related to greater readiness for making behaviour change at the time of pump initiation.
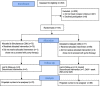
Methods/design: The CGM TIME Trial is a multicenter randomized controlled trial. Eligible children and adolescents (5-18 years) with established type 1 diabetes were randomized to simultaneous initiation of pump (Medtronic Veo©) and CGM (Enlite©) or to standard pump therapy with delayed CGM introduction. Primary outcomes are CGM adherence and hemoglobin A1C at 6 and 12 months post pump initiation. Secondary outcomes include glycemic variability, stage of readiness, and other patient-reported outcomes with follow-up to 24 months. 144 (95%) of the 152 eligible patients were enrolled and randomized. Allowing for 10% withdrawals, this will provide 93% power to detect a between group difference in CGM adherence and 86% power to detect a between group difference in hemoglobin A1C. Baseline characteristics were similar between the treatment groups. Analysis of 12 month primary outcomes will begin in September 2014.
Discussion: The CGM TIME Trial is the first study to examine the relationship between timing of CGM initiation, readiness for behaviour change, and subsequent CGM adherence in pump naïve children and adolescents. Its findings will advance our understanding of when and how to initiate CGM in children and adolescents with type 1 diabetes.
Trial registration: ClinicalTrial.gov NCT01295788. Registered 14 February 2011.
Figures
References
-
- Pickup JC, Freeman SC, Sutton AJ. Glycaemic control in type 1 diabetes during real time continuous glucose monitoring compared with self monitoring of blood glucose: meta-analysis of randomised controlled trials using individual patient data. BMJ. 2011;343:d3805. doi: 10.1136/bmj.d3805. - DOI - PMC - PubMed
-
- The JDRF Continuous Glucose Monitoring Study Group. Continuous glucose monitoring and intensive treatment of type 1 diabetes. N Engl J Med. 2008;359(14):1464–1476. - PubMed
-
- Mauras N, Beck R, Xing D, Ruedy K, Buckingham B, Tansey M, White NH, Weinzimer SA, Tamborlane W, Kollman C. The Diabetes Research in Children Network (DirecNet) Study Group. A randomized clinical trial to assess the efficacy and safety of real-time continuous glucose monitoring in the management of type 1 diabetes in young children aged 4 to <10 years. Diabetes Care. 2012;35(2):204–210. doi: 10.2337/dc11-1746. - DOI - PMC - PubMed
-
- Battelino T, Conget I, Olsen B, Schutz-Fuhrmann I, Hommel E, Hoogma R, Schierloh U, Sulli N, Bolinder J. the SWITCH Study Group. The use and efficacy of continuous glucose monitoring in type 1 diabetes treated with insulin pump therapy: a randomised controlled trial. Diabetologia. 2012;55(12):3155–3162. doi: 10.1007/s00125-012-2708-9. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical