Autophagy defends pancreatic β cells from human islet amyloid polypeptide-induced toxicity
- PMID: 25036708
- PMCID: PMC4109537
- DOI: 10.1172/JCI71981
Autophagy defends pancreatic β cells from human islet amyloid polypeptide-induced toxicity
Abstract
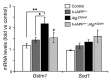
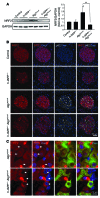
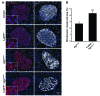
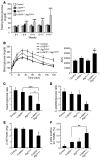
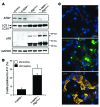
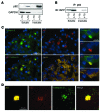
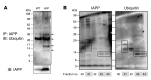
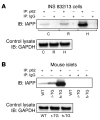
Type 2 diabetes (T2D) is characterized by a deficiency in β cell mass, increased β cell apoptosis, and extracellular accumulation of islet amyloid derived from islet amyloid polypeptide (IAPP), which β cells coexpress with insulin. IAPP expression is increased in the context of insulin resistance, the major risk factor for developing T2D. Human IAPP is potentially toxic, especially as membrane-permeant oligomers, which have been observed to accumulate within β cells of patients with T2D and rodents expressing human IAPP. Here, we determined that β cell IAPP content is regulated by autophagy through p62-dependent lysosomal degradation. Induction of high levels of human IAPP in mouse β cells resulted in accumulation of this amyloidogenic protein as relatively inert fibrils within cytosolic p62-positive inclusions, which temporarily averts formation of toxic oligomers. Mice hemizygous for transgenic expression of human IAPP did not develop diabetes; however, loss of β cell-specific autophagy in these animals induced diabetes, which was attributable to accumulation of toxic human IAPP oligomers and loss of β cell mass. In human IAPP-expressing mice that lack β cell autophagy, increased oxidative damage and loss of an antioxidant-protective pathway appeared to contribute to increased β cell apoptosis. These findings indicate that autophagy/lysosomal degradation defends β cells against proteotoxicity induced by oligomerization-prone human IAPP.
Figures
Comment in
-
Islet amyloid and type 2 diabetes: overproduction or inadequate clearance and detoxification?J Clin Invest. 2014 Aug;124(8):3292-4. doi: 10.1172/JCI77506. Epub 2014 Jul 18. J Clin Invest. 2014. PMID: 25036704 Free PMC article.
-
Diabetes: Protective role of autophagy in pancreatic β cells.Nat Rev Endocrinol. 2014 Oct;10(10):575. doi: 10.1038/nrendo.2014.144. Epub 2014 Aug 12. Nat Rev Endocrinol. 2014. PMID: 25112233 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

