Network modulation following sham surgery in Parkinson's disease
- PMID: 25036712
- PMCID: PMC4109544
- DOI: 10.1172/JCI75073
Network modulation following sham surgery in Parkinson's disease
Abstract
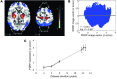
Patient responses to placebo and sham effects are a major obstacle to the development of therapies for brain disorders, including Parkinson's disease (PD). Here, we used functional brain imaging and network analysis to study the circuitry underlying placebo effects in PD subjects randomized to sham surgery as part of a double-blind gene therapy trial. Metabolic imaging was performed prior to randomization, then again at 6 and 12 months after sham surgery. In this cohort, the sham response was associated with the expression of a distinct cerebello-limbic circuit. The expression of this network increased consistently in patients blinded to treatment and correlated with independent clinical ratings. Once patients were unblinded, network expression declined toward baseline levels. Analogous network alterations were not seen with open-label levodopa treatment or during disease progression. Furthermore, sham outcomes in blinded patients correlated with baseline network expression, suggesting the potential use of this quantitative measure to identify "sham-susceptible" subjects before randomization. Indeed, Monte Carlo simulations revealed that a priori exclusion of such individuals substantially lowers the number of randomized participants needed to demonstrate treatment efficacy. Individualized subject selection based on a predetermined network criterion may therefore limit the need for sham interventions in future clinical trials.
Figures





Comment in
-
A brain network response to sham surgery.J Clin Invest. 2014 Aug;124(8):3285-8. doi: 10.1172/JCI77193. Epub 2014 Jul 18. J Clin Invest. 2014. PMID: 25036701 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

