Inducible nitric oxide synthase in neutrophils and endothelium contributes to ischemic brain injury in mice
- PMID: 25038255
- PMCID: PMC4147670
- DOI: 10.4049/jimmunol.1400918
Inducible nitric oxide synthase in neutrophils and endothelium contributes to ischemic brain injury in mice
Abstract
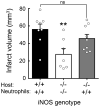
NO produced by inducible NO synthase (iNOS) contributes to ischemic brain injury, but the cell types expressing iNOS and mediating tissue damage have not been elucidated. To examine the relative contribution of iNOS in resident brain cells and peripheral leukocytes infiltrating the ischemic brain, we used bone marrow (BM) chimeric mice in which the middle cerebral artery was occluded and infarct volume was determined 3 d later. iNOS(-/-) mice engrafted with iNOS(+/+) BM exhibited larger infarcts (44 ± 2 mm(3); n = 13; mean ± SE) compared with autologous transplanted iNOS(-/-) mice (24 ± 3 mm(3); n = 10; p < 0.01), implicating blood-borne leukocytes in the damage. Furthermore, iNOS(+/+) mice transplanted with iNOS(-/-) BM had large infarcts (39 ± 6 mm(3); n = 13), similar to those of autologous transplanted iNOS(+/+) mice (39 ± 4 mm(3); n = 14), indicating the resident brain cells also play a role. Flow cytometry and cell sorting revealed that iNOS is highly expressed in neutrophils and endothelium but not microglia. Surprisingly, postischemic iNOS expression was enhanced in the endothelium of iNOS(+/+) mice transplanted with iNOS(-/-) BM and in leukocytes of iNOS(-/-) mice with iNOS(+/+) BM, suggesting that endothelial iNOS suppresses iNOS expression in leukocytes and vice versa. To provide independent evidence that neutrophils mediate brain injury, neutrophils were isolated and transferred to mice 24 h after stroke. Consistent with the result in chimeric mice, transfer of iNOS(+/+), but not iNOS(-/-), neutrophils into iNOS(-/-) mice increased infarct volume. The findings establish that iNOS in both neutrophils and endothelium mediates tissue damage and identify these cell types as putative therapeutic targets for stroke injury.
Copyright © 2014 by The American Association of Immunologists, Inc.
Figures



References
-
- Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ, Dai S, Ford ES, Fox CS, Franco S, Fullerton HJ, Gillespie C, Hailpern SM, Heit JA, Howard VJ, Huffman MD, Judd SE, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Mackey RH, Magid DJ, Marcus GM, Marelli A, Matchar DB, McGuire DK, Mohler ER, Moy CS, Mussolino ME, Neumar RW, Nichol G, Pandey DK, Paynter NP, Reeves MJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Wong ND, Woo D, Turner MB American Heart Association Statistics Committee and Stroke Statistics Subcommittee. . Heart disease and stroke statistics--2014 update: a report from the American Heart Association. Circulation. 2014;129:e28–e292. - PMC - PubMed
-
- Fonarow GC, Smith EE, Saver JL, Reeves MJ, Bhatt DL, Grau-Sepulveda MV, Olson DM, Hernandez AF, Peterson ED, Schwamm LH. Timeliness of Tissue-Type Plasminogen Activator Therapy in Acute Ischemic Stroke: Patient Characteristics, Hospital Factors, and Outcomes Associated With Door-to-Needle Times Within 60 Minutes. Circulation. 2011;123:750–758. - PubMed
-
- Shichita T, Ago T, Kamouchi M, Kitazono T, Yoshimura A, Ooboshi H. Novel therapeutic strategies targeting innate immune responses and early inflammation after stroke. J Neurochem. 2012;123:29–38. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

