Vector design Tour de Force: integrating combinatorial and rational approaches to derive novel adeno-associated virus variants
- PMID: 25048217
- PMCID: PMC4429732
- DOI: 10.1038/mt.2014.139
Vector design Tour de Force: integrating combinatorial and rational approaches to derive novel adeno-associated virus variants
Abstract
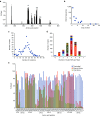
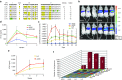
Methodologies to improve existing adeno-associated virus (AAV) vectors for gene therapy include either rational approaches or directed evolution to derive capsid variants characterized by superior transduction efficiencies in targeted tissues. Here, we integrated both approaches in one unified design strategy of "virtual family shuffling" to derive a combinatorial capsid library whereby only variable regions on the surface of the capsid are modified. Individual sublibraries were first assembled in order to preselect compatible amino acid residues within restricted surface-exposed regions to minimize the generation of dead-end variants. Subsequently, the successful families were interbred to derive a combined library of ~8 × 10(5) complexity. Next-generation sequencing of the packaged viral DNA revealed capsid surface areas susceptible to directed evolution, thus providing guidance for future designs. We demonstrated the utility of the library by deriving an AAV2-based vector characterized by a 20-fold higher transduction efficiency in murine liver, now equivalent to that of AAV8.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

