Alteration of vascular reactivity in heart failure: role of phosphodiesterases 3 and 4
- PMID: 25048877
- PMCID: PMC4294045
- DOI: 10.1111/bph.12853
Alteration of vascular reactivity in heart failure: role of phosphodiesterases 3 and 4
Abstract
Background and purpose: This study examined the role of the main vascular cAMP-hydrolysing phosphodiesterases (cAMP-PDE) in the regulation of basal vascular tone and relaxation of rat aorta mediated by β-adrenoceptors, following heart failure (HF).
Experimental approach: Twenty-two weeks after proximal aortic stenosis, to induce HF, or SHAM surgery in rats, we evaluated the expression, activity and function of cAMP-PDE in the descending thoracic aorta.
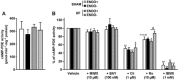
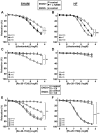
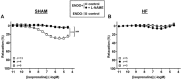
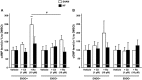
Key results: HF rat aortas exhibited signs of endothelial dysfunction, with alterations of the NO pathway, and alteration of PDE3 and PDE4 subtype expression, without changing total aortic cAMP-hydrolytic activity and PDE1, PDE3 and PDE4 activities. Vascular reactivity experiments using PDE inhibitors showed that PDE3 and PDE4 controlled the level of PGF2α -stimulated contraction in SHAM aorta. PDE3 function was partially inhibited by endothelial NO, whereas PDE4 function required a functional endothelium and was under the negative control of PDE3. In HF, PDE3 function was preserved, but its regulation by endothelial NO was altered. PDE4 function was abolished and restored by PDE3 inhibition. In PGF2α -precontracted arteries, β-adrenoceptor stimulation-induced relaxation in SHAM aorta, which was abolished in the absence of functional endothelium, as well as in HF aortas, but restored after PDE3 inhibition in all unresponsive arteries.
Conclusions and implications: Our study underlines the key role of the endothelium in controlling the contribution of smooth muscle PDE to contractile function. In HF, endothelial dysfunction had a major effect on PDE3 function and PDE3 inhibition restored a functional relaxation to β-adrenoceptor stimulation.
© 2014 The British Pharmacological Society.
Figures




Similar articles
-
Phosphodiesterases type 2, 3 and 4 promote vascular tone in mesenteric arteries from rats with heart failure.Eur J Pharmacol. 2023 Apr 5;944:175562. doi: 10.1016/j.ejphar.2023.175562. Epub 2023 Feb 1. Eur J Pharmacol. 2023. PMID: 36736940
-
Contribution of BKCa channels to vascular tone regulation by PDE3 and PDE4 is lost in heart failure.Cardiovasc Res. 2019 Jan 1;115(1):130-144. doi: 10.1093/cvr/cvy161. Cardiovasc Res. 2019. PMID: 29939224
-
β-Adrenergic cAMP signals are predominantly regulated by phosphodiesterase type 4 in cultured adult rat aortic smooth muscle cells.PLoS One. 2012;7(10):e47826. doi: 10.1371/journal.pone.0047826. Epub 2012 Oct 18. PLoS One. 2012. PMID: 23094097 Free PMC article.
-
[Cyclic nucleotide phosphodiesterases: role in the heart and therapeutic perspectives].Biol Aujourdhui. 2016;210(3):127-138. doi: 10.1051/jbio/2016019. Epub 2016 Nov 4. Biol Aujourdhui. 2016. PMID: 27813474 Review. French.
-
Dual PDE3/4 inhibitors as therapeutic agents for chronic obstructive pulmonary disease.Br J Pharmacol. 2009 Jul;157(6):892-906. doi: 10.1111/j.1476-5381.2009.00170.x. Epub 2009 Jun 5. Br J Pharmacol. 2009. PMID: 19508401 Free PMC article. Review.
Cited by
-
A cardiac mitochondrial cAMP signaling pathway regulates calcium accumulation, permeability transition and cell death.Cell Death Dis. 2016 Apr 21;7(4):e2198. doi: 10.1038/cddis.2016.106. Cell Death Dis. 2016. PMID: 27100892 Free PMC article.
-
Activation of bitter taste receptors (tas2rs) relaxes detrusor smooth muscle and suppresses overactive bladder symptoms.Oncotarget. 2016 Apr 19;7(16):21156-67. doi: 10.18632/oncotarget.8549. Oncotarget. 2016. PMID: 27056888 Free PMC article.
-
The Use of Wire Myography to Investigate Vascular Tone and Function.Methods Mol Biol. 2022;2419:361-376. doi: 10.1007/978-1-0716-1924-7_23. Methods Mol Biol. 2022. PMID: 35237977
-
The Complexity and Multiplicity of the Specific cAMP Phosphodiesterase Family: PDE4, Open New Adapted Therapeutic Approaches.Int J Mol Sci. 2022 Sep 13;23(18):10616. doi: 10.3390/ijms231810616. Int J Mol Sci. 2022. PMID: 36142518 Free PMC article. Review.
-
PDE-Mediated Cyclic Nucleotide Compartmentation in Vascular Smooth Muscle Cells: From Basic to a Clinical Perspective.J Cardiovasc Dev Dis. 2021 Dec 22;9(1):4. doi: 10.3390/jcdd9010004. J Cardiovasc Dev Dis. 2021. PMID: 35050214 Free PMC article. Review.
References
-
- Boess FG, Hendrix M, van der Staay FJ, Erb C, Schreiber R, van Staveren W, et al. Inhibition of phosphodiesterase 2 increases neuronal cGMP, synaptic plasticity and memory performance. Neuropharmacology. 2004;47:1081–1092. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

