Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death
- PMID: 25071979
- PMCID: PMC4106151
- DOI: 10.4161/onci.28518
Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death
Abstract
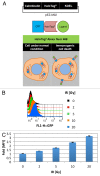
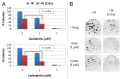
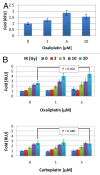
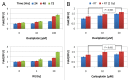
Established tumors are typified by an immunosuppresive microenvironment. Countering this naturally occurring phenomenon, emerging evidence suggests that radiation promotes a proimmunogenic milieu within the tumor capable of stimulating host cancer-specific immune responses. Three cryptic immunogenic components of cytotoxic-agent induced cell death-namely, calreticulin cell surface exposure, the release of high mobility group box 1 (HMGB1) protein, and the liberation of ATP-have been previously shown to be critical for dendritic cell (DC) activation and effector T-cell priming. Thus, these immune-mobilizing components commonly presage tumor rejection in response to treatment. We initially set out to address the hypothesis that radiation-induced immunogenic cell death (ICD) is dose-dependent. Next, we hypothesized that radiation would enhance chemotherapy-induced ICD when given concomitantly, as suggested by the favorable clinical outcomes observed in response to analogous concurrent chemoradiation regimens. Thus, we designed an in vitro assay to examine the 3 hallmark features of ICD at clinically relevant doses of radiation. We then tested the immunogenic-death inducing effects of radiation combined with carboplatin or paclitaxel, focusing on these combinations to mimic chemoradiation regimens actually used in clinical trials of early stage triple negative [NCT0128953/NYU-10-01969] and locally advanced [NYU-06209] breast cancer patients, respectively. Despite the obvious limitations of an in vitro model, radiotherapy produced both a dose-dependent induction and chemotherapeutic enhancement of ICD. These findings provide preliminary evidence that ICD stimulated by either high-dose radiotherapy alone, or concurrent chemoradiation regimens, may contribute to the establishment of a peritumoral proimmunogenic milieu.
Keywords: Immunogenic cell death; carboplatin; ionizing radiation; oxaliplatin; paclitaxel.
Figures




References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
