Efficacy and safety of the RTS,S/AS01 malaria vaccine during 18 months after vaccination: a phase 3 randomized, controlled trial in children and young infants at 11 African sites
- PMID: 25072396
- PMCID: PMC4114488
- DOI: 10.1371/journal.pmed.1001685
Efficacy and safety of the RTS,S/AS01 malaria vaccine during 18 months after vaccination: a phase 3 randomized, controlled trial in children and young infants at 11 African sites
Abstract
Background: A malaria vaccine could be an important addition to current control strategies. We report the safety and vaccine efficacy (VE) of the RTS,S/AS01 vaccine during 18 mo following vaccination at 11 African sites with varying malaria transmission.
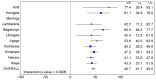
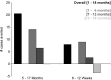
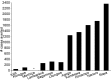
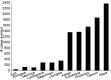
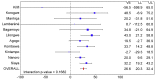
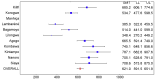
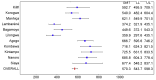
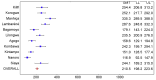
Methods and findings: 6,537 infants aged 6-12 wk and 8,923 children aged 5-17 mo were randomized to receive three doses of RTS,S/AS01 or comparator vaccine. VE against clinical malaria in children during the 18 mo after vaccine dose 3 (per protocol) was 46% (95% CI 42% to 50%) (range 40% to 77%; VE, p<0.01 across all sites). VE during the 20 mo after vaccine dose 1 (intention to treat [ITT]) was 45% (95% CI 41% to 49%). VE against severe malaria, malaria hospitalization, and all-cause hospitalization was 34% (95% CI 15% to 48%), 41% (95% CI 30% to 50%), and 19% (95% CI 11% to 27%), respectively (ITT). VE against clinical malaria in infants was 27% (95% CI 20% to 32%, per protocol; 27% [95% CI 21% to 33%], ITT), with no significant protection against severe malaria, malaria hospitalization, or all-cause hospitalization. Post-vaccination anti-circumsporozoite antibody geometric mean titer varied from 348 to 787 EU/ml across sites in children and from 117 to 335 EU/ml in infants (per protocol). VE waned over time in both age categories (Schoenfeld residuals p<0.001). The number of clinical and severe malaria cases averted per 1,000 children vaccinated ranged across sites from 37 to 2,365 and from -1 to 49, respectively; corresponding ranges among infants were -10 to 1,402 and -13 to 37, respectively (ITT). Meningitis was reported as a serious adverse event in 16/5,949 and 1/2,974 children and in 9/4,358 and 3/2,179 infants in the RTS,S/AS01 and control groups, respectively.
Conclusions: RTS,S/AS01 prevented many cases of clinical and severe malaria over the 18 mo after vaccine dose 3, with the highest impact in areas with the greatest malaria incidence. VE was higher in children than in infants, but even at modest levels of VE, the number of malaria cases averted was substantial. RTS,S/AS01 could be an important addition to current malaria control in Africa.
Trial registration: www.ClinicalTrials.gov NCT00866619 Please see later in the article for the Editors' Summary.
Conflict of interest statement
The trial was sponsored by GlaxoSmithKline Biologicals SA (GSK), the vaccine developer and manufacturer, and funded by both GSK Biologicals SA and the PATH Malaria Vaccine Initiative (MVI). All centers declare receiving a grant from MVI for running the trial. Author travel and accommodation related to this trial were financed by MVI. GlaxoSmithKline Biologicals SA received a grant from MVI to run the trial. MVI received a grant from the Bill & Melinda Gates Foundation to run this trial and to compensate MVI authors for trial-related travel. Additional conflicts of interest are as follows: JJA and PAl declare that their institutions received grant from the Catalan government and from the International Agency for Development and Cooperation. NA, CO, and KO declare that their institutions received a grant from the Malaria Clinical Trial Alliance. PB, SD, BG, CK, PL, CMai, GMwam, BO, and LO declare that their institution has received grants from MVI for other malaria studies. KM declares that his institution received a grant from the Wellcome Trust and that he received support from USAID and the Bill & Melinda Gates Foundation to participate in a scientific advisory group on malaria. MML declares that she received non-financial support from the WHO and the Biomérieux Foundation. PN declares that she received financial support from GSK to present the results of the study at ASTMH congress in 2012. LO declares that he received support from GSK to carry out clinical and epidemiological studies. JSa has received (for the Center) some GlaxoSmithKline group of companies' consultancy fees for other studies. MTa is a board member of the Optimus Foundation, and his institution is reimbursed for his activities on the scientific advisory board of the Novartis Institute for Tropical Diseases. He also has received for his institution other grants from MVI and from the Bill & Melinda Gates Foundation, and travel reimbursements from MVI and Sanaria corp. All GSK Vaccines authors are, or were at the time of the study, employed by the GlaxoSmithKline group of companies. JC now works as an independent consultant for GSK Vaccines. WRB, JC, EJ, DLa, OOA, JV, AL, and MLi have shares/stock options in the GlaxoSmithKline group of companies. JC and WRB declare that they are named inventors on patents for which the rights have been assigned to GlaxoSmithKline group of companies. DK, DLe, and BS are employees at PATH-MVI. DSc is employed by the London School of Hygiene & Tropical Medicine, and his consultancy activities for MVI are funded as a grant to the LSHTM by MVI. DK holds stock or stock options from Merck, Sharpe & Dome.
Figures








Comment in
-
Phase 3 trial with the RTS,S/AS01 malaria vaccine shows protection against clinical and severe malaria in infants and children in Africa.Evid Based Med. 2015 Feb;20(1):9. doi: 10.1136/ebmed-2014-110089. Epub 2014 Sep 16. Evid Based Med. 2015. PMID: 25228673 No abstract available.
References
-
- Murray CJ, Rosenfeld LC, Lim SS, Andrews KG, Foreman KJ, et al. (2012) Global malaria mortality between 1980 and 2010: a systematic analysis. Lancet 379: 413–431. - PubMed
-
- World Health Organization (2012) World malaria report 2012. Geneva: World Health Organization.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

