Regulation of human growth hormone receptor expression by microRNAs
- PMID: 25073105
- PMCID: PMC5414794
- DOI: 10.1210/me.2014-1183
Regulation of human growth hormone receptor expression by microRNAs
Abstract
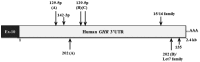
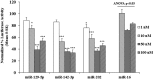
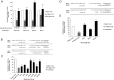
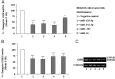
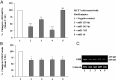
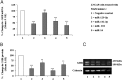
Human GH binds to its receptor (GHR) on target cells and activates multiple intracellular pathways, leading to changes in gene expression, differentiation, and metabolism. GHR deficiency is associated with growth and metabolic disorders whereas increased GHR expression has been reported in certain cancers, suggesting that the GHR gene requires tight controls. Several regulatory mechanisms have been found within its 5'-untranslated region (UTR) promoter and coding regions. However, the 3'-UTR has not been previously examined. MicroRNAs (miRNAs) are small (19-22 nucleotides) noncoding RNAs that downregulate gene expression mainly through targeting the 3'-UTR of mRNAs and enhancing their degradation or inhibiting translation. In the present study, we investigated whether miRNAs regulate GHR expression. To define putative miRNA binding sites in the GHR 3'-UTR, we used multiple in silico prediction tools, analyzed conservation across species and the presence of parallel sites in GH/IGF axis-related genes, and searched for reports linking miRNAs to GHR-related physiological or pathophysiological activities. To test prioritized sites, we cotransfected a wild-type GHR 3'-UTR luciferase reporter vector as well as miRNA binding site mutants into HEK293 cells with miRNA mimics. Furthermore, we tested whether the miRNAs altered endogenous GHR mRNA and protein levels in HEK293 cells and in 2 cancer cell lines (MCF7 and LNCaP). Our experiments have identified miRNA (miR)-129-5p, miR-142-3p, miR-202, and miR-16 as potent inhibitors of human GHR expression in normal (HEK293) and cancer (MCF7 and LNCaP) cells. This study paves the way for the development of miRNA inhibitors as therapeutic agents in GH/GHR-related pathophysiologies, including cancer.
Figures
Similar articles
-
Integrated analysis of noncoding RNAs and mRNAs reveals their potential roles in the biological activities of the growth hormone receptor.Growth Horm IGF Res. 2016 Aug;29:11-20. doi: 10.1016/j.ghir.2016.03.003. Epub 2016 Apr 3. Growth Horm IGF Res. 2016. PMID: 27064376
-
miR-150, p53 protein and relevant miRNAs consist of a regulatory network in NSCLC tumorigenesis.Oncol Rep. 2013 Jul;30(1):492-8. doi: 10.3892/or.2013.2453. Epub 2013 May 13. Oncol Rep. 2013. PMID: 23670238
-
SOX9 is a target of miR-134-3p and miR-224-3p in breast cancer cell lines.Mol Cell Biochem. 2023 Feb;478(2):305-315. doi: 10.1007/s11010-022-04507-z. Epub 2022 Jul 2. Mol Cell Biochem. 2023. PMID: 35779228 Free PMC article.
-
Regulation of the MIR155 host gene in physiological and pathological processes.Gene. 2013 Dec 10;532(1):1-12. doi: 10.1016/j.gene.2012.12.009. Epub 2012 Dec 14. Gene. 2013. PMID: 23246696 Review.
-
Transcriptional regulation of nuclear miRNAs in tumorigenesis (Review).Int J Mol Med. 2022 Jul;50(1):92. doi: 10.3892/ijmm.2022.5148. Epub 2022 May 20. Int J Mol Med. 2022. PMID: 35593304 Review.
Cited by
-
Fetal malnutrition-induced catch up failure is caused by elevated levels of miR-322 in rats.Sci Rep. 2020 Jan 28;10(1):1339. doi: 10.1038/s41598-020-58392-x. Sci Rep. 2020. PMID: 31992823 Free PMC article.
-
MiR-16 mediates trastuzumab and lapatinib response in ErbB-2-positive breast and gastric cancer via its novel targets CCNJ and FUBP1.Oncogene. 2016 Dec 1;35(48):6189-6202. doi: 10.1038/onc.2016.151. Epub 2016 May 9. Oncogene. 2016. PMID: 27157613 Free PMC article.
-
Specific miRNAs Change After 3 Months of GH treatment and Contribute to Explain the Growth Response After 12 Months.Front Endocrinol (Lausanne). 2022 Jun 22;13:896640. doi: 10.3389/fendo.2022.896640. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35813630 Free PMC article.
-
Effects of miR-129-3p on biological functions of prostate cancer cells through targeted regulation of Smad3.Oncol Lett. 2020 Feb;19(2):1195-1202. doi: 10.3892/ol.2019.11216. Epub 2019 Dec 13. Oncol Lett. 2020. PMID: 31966049 Free PMC article.
-
The Metabolism Reprogramming of microRNA Let-7-Mediated Glycolysis Contributes to Autophagy and Tumor Progression.Int J Mol Sci. 2021 Dec 22;23(1):113. doi: 10.3390/ijms23010113. Int J Mol Sci. 2021. PMID: 35008539 Free PMC article. Review.
References
-
- Veldhuis JD, Roemmich JN, Richmond EJ, et al. . Endocrine control of body composition in infancy, childhood, and puberty. Endocr Rev. 2005;26(1):114–146. - PubMed
-
- Lichanska AM, Waters MJ. New insights into growth hormone receptor function and clinical implications. Horm Res. 2008;69(3):138–145. - PubMed
-
- Lichanska AM, Waters MJ. How growth hormone controls growth, obesity and sexual dimorphism. Trends Genet. 2008;24(1):41–47. - PubMed
-
- Brooks AJ, Waters MJ. The growth hormone receptor: mechanism of activation and clinical implications. Nat Rev Endocrinol. 2010;6(9):515–525. - PubMed
-
- Savage MO, Attie KM, David A, Metherell LA, Clark AJ, Camacho-Hubner C. Endocrine assessment, molecular characterization and treatment of growth hormone insensitivity disorders. Nat Clin Pract Endocrinol Metab. 2006;2(7):395–407. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

