Computer simulations reveal complex distribution of haemodynamic forces in a mouse retina model of angiogenesis
- PMID: 25079871
- PMCID: PMC4233731
- DOI: 10.1098/rsif.2014.0543
Computer simulations reveal complex distribution of haemodynamic forces in a mouse retina model of angiogenesis
Abstract
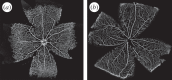
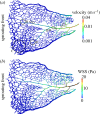
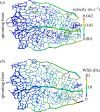
There is currently limited understanding of the role played by haemodynamic forces on the processes governing vascular development. One of many obstacles to be overcome is being able to measure those forces, at the required resolution level, on vessels only a few micrometres thick. In this paper, we present an in silico method for the computation of the haemodynamic forces experienced by murine retinal vasculature (a widely used vascular development animal model) beyond what is measurable experimentally. Our results show that it is possible to reconstruct high-resolution three-dimensional geometrical models directly from samples of retinal vasculature and that the lattice-Boltzmann algorithm can be used to obtain accurate estimates of the haemodynamics in these domains. We generate flow models from samples obtained at postnatal days (P) 5 and 6. Our simulations show important differences between the flow patterns recovered in both cases, including observations of regression occurring in areas where wall shear stress (WSS) gradients exist. We propose two possible mechanisms to account for the observed increase in velocity and WSS between P5 and P6: (i) the measured reduction in typical vessel diameter between both time points and (ii) the reduction in network density triggered by the pruning process. The methodology developed herein is applicable to other biomedical domains where microvasculature can be imaged but experimental flow measurements are unavailable or difficult to obtain.
Keywords: angiogenesis; blood flow; lattice-Boltzmann; mouse; retina; shear stress.
Figures


 and lattice-Boltzmann (LB) relaxation time
and lattice-Boltzmann (LB) relaxation time  . For
. For  , the total error is kept below 3% even for cylinders with just three lattice sites across. These results confirm the suitability of the LB algorithm for the simulation of flow in sparse geometries and porous media. The lines are a guide to the eye and bear no physical meaning. (Online version in colour.)
, the total error is kept below 3% even for cylinders with just three lattice sites across. These results confirm the suitability of the LB algorithm for the simulation of flow in sparse geometries and porous media. The lines are a guide to the eye and bear no physical meaning. (Online version in colour.)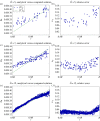
 . Norm of the analytical and computed stress tensors (panels 4a,c,e) and relative error between them (panels 4b,d,f). Results are presented for every lattice site with radius
. Norm of the analytical and computed stress tensors (panels 4a,c,e) and relative error between them (panels 4b,d,f). Results are presented for every lattice site with radius  . Agreement between computed and analytical solution improves with increasing
. Agreement between computed and analytical solution improves with increasing  . These results, with the Bouzidi et al. [50] implementation of the no-slip boundary condition, represent a substantial improvement over the 35% error reported by Stahl et al. [51] with the bounce-back method and
. These results, with the Bouzidi et al. [50] implementation of the no-slip boundary condition, represent a substantial improvement over the 35% error reported by Stahl et al. [51] with the bounce-back method and  . (Online version in colour.)
. (Online version in colour.)
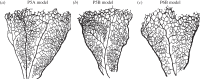



 on simulations with the P6A flow model discretized with Δxi = 1.0, 0.5 and 0.25 μm (diamond-shaped markers). In order to keep the analysis computationally tractable,
on simulations with the P6A flow model discretized with Δxi = 1.0, 0.5 and 0.25 μm (diamond-shaped markers). In order to keep the analysis computationally tractable,  is computed with results obtained at the lattice sites located on the z = 0 plane only (this is the same subset of the results presented in figure 9). The lines are guides to the eye showing first-order (dashed) and second-order (solid) convergence. (Online version in colour.)
is computed with results obtained at the lattice sites located on the z = 0 plane only (this is the same subset of the results presented in figure 9). The lines are guides to the eye showing first-order (dashed) and second-order (solid) convergence. (Online version in colour.)
References
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
