Endocytosis of viruses and bacteria
- PMID: 25085912
- PMCID: PMC4107984
- DOI: 10.1101/cshperspect.a016972
Endocytosis of viruses and bacteria
Abstract
Of the many pathogens that infect humans and animals, a large number use cells of the host organism as protected sites for replication. To reach the relevant intracellular compartments, they take advantage of the endocytosis machinery and exploit the network of endocytic organelles for penetration into the cytosol or as sites of replication. In this review, we discuss the endocytic entry processes used by viruses and bacteria and compare the strategies used by these dissimilar classes of pathogens.
Copyright © 2014 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures

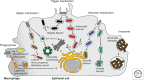

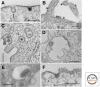
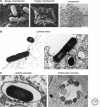
References
-
- Backert S, Meyer TF 2006. Type IV secretion systems and their effectors in bacterial pathogenesis. Curr Opin Microbiol 9: 207–217 - PubMed
-
- Bakowski MA, Braun V, Lam GY, Yeung T, Heo WD, Meyer T, Finlay BB, Grinstein S, Brumell JH 2010. The phosphoinositide phosphatase SopB manipulates membrane surface charge and trafficking of the Salomnella-containg vacuole. Cell Host Microbe 7: 453–462 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
