Vildagliptin stimulates endothelial cell network formation and ischemia-induced revascularization via an endothelial nitric-oxide synthase-dependent mechanism
- PMID: 25100725
- PMCID: PMC4175356
- DOI: 10.1074/jbc.M114.557835
Vildagliptin stimulates endothelial cell network formation and ischemia-induced revascularization via an endothelial nitric-oxide synthase-dependent mechanism
Abstract
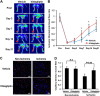
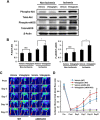
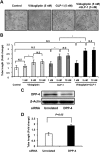
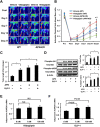
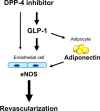
Dipeptidyl peptidase-4 inhibitors are known to lower glucose levels and are also beneficial in the management of cardiovascular disease. Here, we investigated whether a dipeptidyl peptidase-4 inhibitor, vildagliptin, modulates endothelial cell network formation and revascularization processes in vitro and in vivo. Treatment with vildagliptin enhanced blood flow recovery and capillary density in the ischemic limbs of wild-type mice, with accompanying increases in phosphorylation of Akt and endothelial nitric-oxide synthase (eNOS). In contrast to wild-type mice, treatment with vildagliptin did not improve blood flow in ischemic muscles of eNOS-deficient mice. Treatment with vildagliptin increased the levels of glucagon-like peptide-1 (GLP-1) and adiponectin, which have protective effects on the vasculature. Both vildagliptin and GLP-1 increased the differentiation of cultured human umbilical vein endothelial cells (HUVECs) into vascular-like structures, although vildagliptin was less effective than GLP-1. GLP-1 and vildagliptin also stimulated the phosphorylation of Akt and eNOS in HUVECs. Pretreatment with a PI3 kinase or NOS inhibitor blocked the stimulatory effects of both vildagliptin and GLP-1 on HUVEC differentiation. Furthermore, treatment with vildagliptin only partially increased the limb flow of ischemic muscle in adiponectin-deficient mice in vivo. GLP-1, but not vildagliptin, significantly increased adiponectin expression in differentiated 3T3-L1 adipocytes in vitro. These data indicate that vildagliptin promotes endothelial cell function via eNOS signaling, an effect that may be mediated by both GLP-1-dependent and GLP-1-independent mechanisms. The beneficial activity of GLP-1 for revascularization may also be partially mediated by its ability to increase adiponectin production.
Keywords: Adiponectin; Angiogenesis; Endothelial Cell; Nitric-oxide Synthase; Signaling.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


Similar articles
-
Fat-derived factor omentin stimulates endothelial cell function and ischemia-induced revascularization via endothelial nitric oxide synthase-dependent mechanism.J Biol Chem. 2012 Jan 2;287(1):408-417. doi: 10.1074/jbc.M111.261818. Epub 2011 Nov 11. J Biol Chem. 2012. PMID: 22081609 Free PMC article.
-
Dipeptidyl peptidase 4 inhibitor reduces intimal hyperplasia in rabbit autologous jugular vein graft under poor distal runoff.J Vasc Surg. 2016 May;63(5):1360-70. doi: 10.1016/j.jvs.2014.12.048. Epub 2015 Feb 2. J Vasc Surg. 2016. PMID: 25656593
-
Diallyl Trisulfide Augments Ischemia-Induced Angiogenesis via an Endothelial Nitric Oxide Synthase-Dependent Mechanism.Circ J. 2017 May 25;81(6):870-878. doi: 10.1253/circj.CJ-16-1097. Epub 2017 Feb 17. Circ J. 2017. PMID: 28216514
-
Mechanisms of action of the dipeptidyl peptidase-4 inhibitor vildagliptin in humans.Diabetes Obes Metab. 2011 Sep;13(9):775-83. doi: 10.1111/j.1463-1326.2011.01414.x. Diabetes Obes Metab. 2011. PMID: 21507182 Review.
-
The scientific evidence: vildagliptin and the benefits of islet enhancement.Diabetes Obes Metab. 2009 May;11 Suppl 2:9-17. doi: 10.1111/j.1463-1326.2008.01033.x. Diabetes Obes Metab. 2009. PMID: 19385979 Review.
Cited by
-
Vildagliptin Reduces Stenosis of Injured Carotid Artery in Diabetic Mouse Through Inhibiting Vascular Smooth Muscle Cell Proliferation via ER Stress/NF-κB Pathway.Front Pharmacol. 2019 Feb 25;10:142. doi: 10.3389/fphar.2019.00142. eCollection 2019. Front Pharmacol. 2019. PMID: 30858802 Free PMC article.
-
Vildagliptin, but not glibenclamide, increases circulating endothelial progenitor cell number: a 12-month randomized controlled trial in patients with type 2 diabetes.Cardiovasc Diabetol. 2017 Feb 23;16(1):27. doi: 10.1186/s12933-017-0503-0. Cardiovasc Diabetol. 2017. PMID: 28231835 Free PMC article. Clinical Trial.
-
DPP-4 Inhibitors as Potential Candidates for Antihypertensive Therapy: Improving Vascular Inflammation and Assisting the Action of Traditional Antihypertensive Drugs.Front Immunol. 2019 May 9;10:1050. doi: 10.3389/fimmu.2019.01050. eCollection 2019. Front Immunol. 2019. PMID: 31134095 Free PMC article. Review.
-
Metabolic Syndrome: Updates on Pathophysiology and Management in 2021.Int J Mol Sci. 2022 Jan 12;23(2):786. doi: 10.3390/ijms23020786. Int J Mol Sci. 2022. PMID: 35054972 Free PMC article. Review.
-
Augmentation of RBP4/STRA6 signaling leads to insulin resistance and inflammation and the plausible therapeutic role of vildagliptin and metformin.Mol Biol Rep. 2021 May;48(5):4093-4106. doi: 10.1007/s11033-021-06420-y. Epub 2021 May 26. Mol Biol Rep. 2021. PMID: 34041677
References
-
- Weitz J. I., Byrne J., Clagett G. P., Farkouh M. E., Porter J. M., Sackett D. L., Strandness D. E., Jr., Taylor L. M. (1996) Diagnosis and treatment of chronic arterial insufficiency of the lower extremities: a critical review. Circulation 94, 3026–3049 - PubMed
-
- Wang X. M., Yang Y. J., Wu Y. J. (2013) The emerging role of dipeptidyl peptidase-4 inhibitors in cardiovascular protection: current position and perspectives. Cardiovasc. Drugs Ther. 27, 297–307 - PubMed
-
- Murohara T. (2012) Dipeptidyl peptidase-4 inhibitor: another player for cardiovascular protection. J. Am. Coll. Cardiol. 59, 277–279 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

