Role of GLUT1 in regulation of reactive oxygen species
- PMID: 25101238
- PMCID: PMC4116627
- DOI: 10.1016/j.redox.2014.03.004
Role of GLUT1 in regulation of reactive oxygen species
Abstract
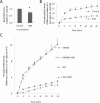
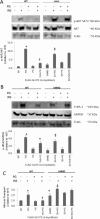
In skeletal muscle cells, GLUT1 is responsible for a large portion of basal uptake of glucose and dehydroascorbic acid, both of which play roles in antioxidant defense. We hypothesized that conditions that would decrease GLUT1-mediated transport would cause increased reactive oxygen species (ROS) levels in L6 myoblasts, while conditions that would increase GLUT1-mediated transport would result in decreased ROS levels. We found that the GLUT1 inhibitors fasentin and phloretin increased the ROS levels induced by antimycin A and the superoxide generator pyrogallol. However, indinavir, which inhibits GLUT4 but not GLUT1, had no effect on ROS levels. Ataxia telangiectasia mutated (ATM) inhibitors and activators, previously shown to inhibit and augment GLUT1-mediated transport, increased and decreased ROS levels, respectively. Mutation of an ATM target site on GLUT1 (GLUT1-S490A) increased ROS levels and prevented the ROS-lowering effect of the ATM activator doxorubicin. In contrast, expression of GLUT1-S490D lowered ROS levels during challenge with pyrogallol, prevented an increase in ROS when ATM was inhibited, and prevented the pyrogallol-induced decrease in insulin signaling and insulin-stimulated glucose transport. Taken together, the data suggest that GLUT1 plays a role in regulation of ROS and could contribute to maintenance of insulin action in the presence of ROS.
Keywords: Antioxidant; Ataxia telangiectasia mutated (ATM); Glucose transport; Insulin resistance; Oxidative stress.
Figures



References
-
- Zembron-Lacny A., Naczk M., Gajewski M., Ostapiuk-Karolczuk J., Dziewiecka H., Kasperska A., Szyszka K. Changes of muscle-derived cytokines in relation to thiol redox status and reactive oxygen and nitrogen species. Physiological Research/Academia Scientiarum Bohemoslovaca. 2010;59:945–951. 20533854 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

