Effect of angiotensin II and small GTPase Ras signaling pathway inhibition on early renal changes in a murine model of obstructive nephropathy
- PMID: 25101263
- PMCID: PMC4101960
- DOI: 10.1155/2014/124902
Effect of angiotensin II and small GTPase Ras signaling pathway inhibition on early renal changes in a murine model of obstructive nephropathy
Abstract
Tubulointerstitial fibrosis is a major feature of chronic kidney disease. Unilateral ureteral obstruction (UUO) in rodents leads to the development of renal tubulointerstitial fibrosis consistent with histopathological changes observed in advanced chronic kidney disease in humans. The purpose of this study was to assess the effect of inhibiting angiotensin II receptors or Ras activation on early renal fibrotic changes induced by UUO. Animals either received angiotensin II or underwent UUO. UUO animals received either losartan, atorvastatin, and farnesyl transferase inhibitor (FTI) L-744,832, or chaetomellic acid A (ChA). Levels of activated Ras, phospho-ERK1/2, phospho-Akt, fibronectin, and α-smooth muscle actin were subsequently quantified in renal tissue by ELISA, Western blot, and/or immunohistochemistry. Our results demonstrate that administration of angiotensin II induces activation of the small GTPase Ras/Erk/Akt signaling system, suggesting an involvement of angiotensin II in the early obstruction-induced activation of renal Ras. Furthermore, upstream inhibition of Ras signalling by blocking either angiotensin AT1 type receptor or by inhibiting Ras prenylation (atorvastatin, FTI o ChA) reduced the activation of the Ras/Erk/Akt signaling system and decreased the early fibrotic response in the obstructed kidney. This study points out that pharmacological inhibition of Ras activation may hold promise as a future strategy in the prevention of renal fibrosis.
Figures
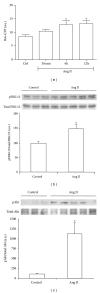
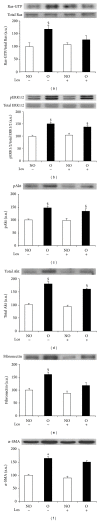


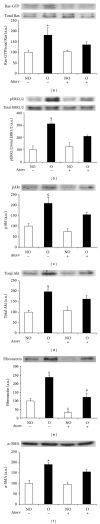
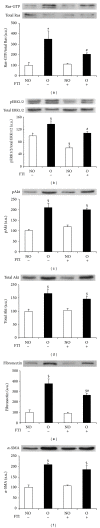
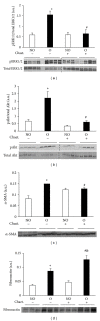
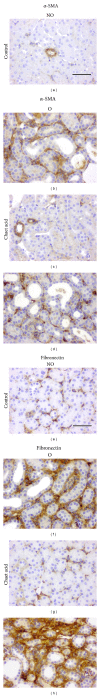
References
-
- Ucero AC, Benito-Martin A, Izquierdo MC, et al. Unilateral ureteral obstruction: beyond obstruction. International Urology and Nephrology. 2014;46(4):765–776. - PubMed
-
- Klahr S, Ishidoya S, Morrissey J. Role of angiotensin II in the tubulointerstitial fibrosis of obstructive nephropathy. American Journal of Kidney Diseases. 1995;26(1):141–146. - PubMed
-
- Pimentel JL, Jr., Montero A, Wang S, Yosipiv I, El-Dahr S, Martinez-Maldonado M. Sequential changes in renal expression of renin-angiotensin system genes in acute unilateral ureteral obstruction. Kidney International. 1995;48(4):1247–1253. - PubMed
-
- Pimentel JL, Jr., Sundell CL, Wang S, Kopp JB, Montero Á, Martínez-Maldonado M. Role of angiotensin II in the expression and regulation of transforming growth factor-β in obstructive nephropathy. Kidney International. 1995;48(4):1233–1246. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

