Multiplex evaluation of influenza neutralizing antibodies with potential applicability to in-field serological studies
- PMID: 25101305
- PMCID: PMC4101955
- DOI: 10.1155/2014/457932
Multiplex evaluation of influenza neutralizing antibodies with potential applicability to in-field serological studies
Abstract
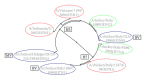
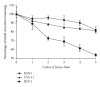
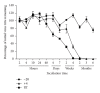
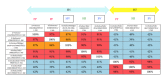
The increased number of outbreaks of H5 and H7 LPAI and HPAI viruses in poultry has major public and animal health implications. The continuous rapid evolution of these subtypes and the emergence of new variants influence the ability to undertake effective surveillance. Retroviral pseudotypes bearing influenza haemagglutinin (HA) and neuraminidase (NA) envelope glycoproteins represent a flexible platform for sensitive, readily standardized influenza serological assays. We describe a multiplex assay for the study of neutralizing antibodies that are directed against both influenza H5 and H7 HA. This assay permits the measurement of neutralizing antibody responses against two antigenically distinct HAs in the same serum/plasma sample thus increasing the amount and quality of serological data that can be acquired from valuable sera. Sera obtained from chickens vaccinated with a monovalent H5N2 vaccine, chickens vaccinated with a bivalent H7N1/H5N9 vaccine, or turkeys naturally infected with an H7N3 virus were evaluated in this assay and the results correlated strongly with data obtained by HI assay. We show that pseudotypes are highly stable under basic cold-chain storage conditions and following multiple rounds of freeze-thaw. We propose that this robust assay may have practical utility for in-field serosurveillance and vaccine studies in resource-limited regions worldwide.
Figures


Similar articles
-
Protection of chickens against highly lethal H5N1 and H7N1 avian influenza viruses with a recombinant fowlpox virus co-expressing H5 haemagglutinin and N1 neuraminidase genes.Avian Pathol. 2003 Feb;32(1):25-32. doi: 10.1080/0307945021000070688. Avian Pathol. 2003. PMID: 12745375 Clinical Trial.
-
Virus-like particles displaying H5, H7, H9 hemagglutinins and N1 neuraminidase elicit protective immunity to heterologous avian influenza viruses in chickens.Virology. 2017 Jan 15;501:176-182. doi: 10.1016/j.virol.2016.12.001. Epub 2016 Dec 6. Virology. 2017. PMID: 27936463 Free PMC article.
-
Inactivated H7 Influenza Virus Vaccines Protect Mice despite Inducing Only Low Levels of Neutralizing Antibodies.J Virol. 2017 Sep 27;91(20):e01202-17. doi: 10.1128/JVI.01202-17. Print 2017 Oct 15. J Virol. 2017. PMID: 28768855 Free PMC article.
-
Epitopes in the HA and NA of H5 and H7 avian influenza viruses that are important for antigenic drift.FEMS Microbiol Rev. 2024 May 8;48(3):fuae014. doi: 10.1093/femsre/fuae014. FEMS Microbiol Rev. 2024. PMID: 38734891 Free PMC article. Review.
-
Development and use of fowlpox vectored vaccines for avian influenza.Ann N Y Acad Sci. 2006 Oct;1081:193-201. doi: 10.1196/annals.1373.023. Ann N Y Acad Sci. 2006. PMID: 17135511 Review.
Cited by
-
Characterisation of SARS-CoV-2 Lentiviral Pseudotypes and Correlation between Pseudotype-Based Neutralisation Assays and Live Virus-Based Micro Neutralisation Assays.Viruses. 2020 Sep 10;12(9):1011. doi: 10.3390/v12091011. Viruses. 2020. PMID: 32927639 Free PMC article.
-
The Optimisation of Pseudotyped Viruses for the Characterisation of Immune Responses to Equine Influenza Virus.Pathogens. 2016 Dec 15;5(4):68. doi: 10.3390/pathogens5040068. Pathogens. 2016. PMID: 27983716 Free PMC article.
-
An Innovative Pseudotypes-Based Enzyme-Linked Lectin Assay for the Measurement of Functional Anti-Neuraminidase Antibodies.PLoS One. 2015 Aug 12;10(8):e0135383. doi: 10.1371/journal.pone.0135383. eCollection 2015. PLoS One. 2015. PMID: 26267900 Free PMC article.
-
Pathological Changes in Natural Infection of Pheasants with Highly Pathogenic Avian Influenza A (H5N8) in Bulgaria.J Vet Res. 2019 Dec 19;63(4):497-502. doi: 10.2478/jvetres-2019-0073. eCollection 2019 Dec. J Vet Res. 2019. PMID: 31934658 Free PMC article.
-
More Than Just Gene Therapy Vectors: Lentiviral Vector Pseudotypes for Serological Investigation.Viruses. 2021 Jan 31;13(2):217. doi: 10.3390/v13020217. Viruses. 2021. PMID: 33572589 Free PMC article. Review.
References
-
- Capua I, Marangon S. Control and prevention of avian influenza in an evolving scenario. Vaccine. 2007;25(30):5645–5652. - PubMed
-
- Röhm C, Horimoto T, Kawaoka Y, Süss J, Webster RG. Do hemagglutinin genes of highly pathogenic avian influenza viruses constitute unique phylogenetic lineages? Virology. 1995;209(2):664–670. - PubMed
-
- Banks J, Speidel ES, Moore E, et al. Changes in the haemagglutinin and the neuraminidase genes prior to the emergence of highly pathogenic H7N1 avian influenza viruses in Italy. Archives of Virology. 2001;146(5):963–973. - PubMed
-
- García M, Crawford JM, Latimer JW, Rivera-Cruz E, Perdue ML. Heterogeneity in the haemagglutinin gene and emergence of the highly pathogenic phenotype among recent H5N2 avian influenza viruses from Mexico. Journal of General Virology. 1996;77, part 7:1493–1504. - PubMed
-
- Capua I, Marangon S, Dalla Pozza M, Santucci U. Vaccination for avian influenza in Italy. Veterinary Record. 2000;147(26):p. 751. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

