Brain-on-a-chip microsystem for investigating traumatic brain injury: Axon diameter and mitochondrial membrane changes play a significant role in axonal response to strain injuries
- PMID: 25101309
- PMCID: PMC4120884
- DOI: 10.1142/S2339547814500095
Brain-on-a-chip microsystem for investigating traumatic brain injury: Axon diameter and mitochondrial membrane changes play a significant role in axonal response to strain injuries
Abstract
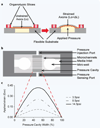
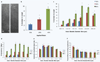
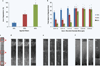
Diffuse axonal injury (DAI) is a devastating consequence of traumatic brain injury, resulting in significant axon and neuronal degeneration. Currently, therapeutic options are limited. Using our brain-on-a-chip device, we evaluated axonal responses to DAI. We observed that axonal diameter plays a significant role in response to strain injury, which correlated to delayed elasticity and inversely correlated to axonal beading and axonal degeneration. When changes in mitochondrial membrane potential (MMP) were monitored an applied strain injury threshold was noted, below which delayed hyperpolarization was observed and above which immediate depolarization occurred. When the NHE-1 inhibitor EIPA was administered before injury, inhibition in both hyperpolarization and depolarization occurred along with axonal degeneration. Therefore, axonal diameter plays a significant role in strain injury and our brain-on-a-chip technology can be used both to understand the biochemical consequences of DAI and screen for potential therapeutic agents.
Figures
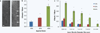
Similar articles
-
A Precise, Controllable in vitro Model for Diffuse Axonal Injury Through Uniaxial Stretch Injury.Front Neurosci. 2019 Oct 17;13:1063. doi: 10.3389/fnins.2019.01063. eCollection 2019. Front Neurosci. 2019. PMID: 31680808 Free PMC article.
-
CLARITY reveals a more protracted temporal course of axon swelling and disconnection than previously described following traumatic brain injury.Brain Pathol. 2019 May;29(3):437-450. doi: 10.1111/bpa.12677. Epub 2018 Dec 27. Brain Pathol. 2019. PMID: 30444552 Free PMC article.
-
Nodal versus Total Axonal Strain and the Role of Cholesterol in Traumatic Brain Injury.J Neurotrauma. 2016 May 1;33(9):859-70. doi: 10.1089/neu.2015.4007. Epub 2015 Sep 22. J Neurotrauma. 2016. PMID: 26393780
-
Current Opportunities for Clinical Monitoring of Axonal Pathology in Traumatic Brain Injury.Front Neurol. 2017 Nov 20;8:599. doi: 10.3389/fneur.2017.00599. eCollection 2017. Front Neurol. 2017. PMID: 29209266 Free PMC article. Review.
-
Current concepts: diffuse axonal injury-associated traumatic brain injury.Arch Phys Med Rehabil. 2001 Oct;82(10):1461-71. doi: 10.1053/apmr.2001.25137. Arch Phys Med Rehabil. 2001. PMID: 11588754 Review.
Cited by
-
Recent progress in translational engineered in vitro models of the central nervous system.Brain. 2020 Dec 5;143(11):3181-3213. doi: 10.1093/brain/awaa268. Brain. 2020. PMID: 33020798 Free PMC article. Review.
-
The microbiota-gut-brain axis and epilepsy from a multidisciplinary perspective: Clinical evidence and technological solutions for improvement of in vitro preclinical models.Bioeng Transl Med. 2022 Feb 25;7(2):e10296. doi: 10.1002/btm2.10296. eCollection 2022 May. Bioeng Transl Med. 2022. PMID: 35600638 Free PMC article. Review.
-
Microdevice Platform for In Vitro Nervous System and Its Disease Model.Bioengineering (Basel). 2017 Sep 13;4(3):77. doi: 10.3390/bioengineering4030077. Bioengineering (Basel). 2017. PMID: 28952555 Free PMC article. Review.
-
A human brain microphysiological system derived from induced pluripotent stem cells to study neurological diseases and toxicity.ALTEX. 2017;34(3):362-376. doi: 10.14573/altex.1609122. Epub 2016 Nov 24. ALTEX. 2017. PMID: 27883356 Free PMC article.
-
In vitro inflammatory multi-cellular model of osteoarthritis.Osteoarthr Cartil Open. 2024 Jan 5;6(1):100432. doi: 10.1016/j.ocarto.2023.100432. eCollection 2024 Mar. Osteoarthr Cartil Open. 2024. PMID: 38288345 Free PMC article.
References
-
- Meythaler JM, Peduzzi JD, Eleftheriou E, Novack TA. Current concepts: Diffuse axonal injury-associated traumatic brain injury. Arch. Phys. Med. Rehabil. 2001;82:1461–1471. - PubMed
-
- Smith DH, Meaney DF. Axonal damage in traumatic brain injury. Neuroscientist. 2000;6:483–495.
-
- Gennarelli TA, et al. Axonal injury in the optic nerve: A model simulating diffuse axonal injury in the brain. J. Neurosurg. 1989;71:244–253. - PubMed
-
- Kilinc D, Gallo G, Barbee KA. Mechanically-induced membrane poration causes axonal beading and localized cytoskeletal damage. Exp. Neurol. 2008;212:422–430. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
