Protective effects of carbon monoxide-releasing molecule-2 on the barrier function of intestinal epithelial cells
- PMID: 25101775
- PMCID: PMC4125175
- DOI: 10.1371/journal.pone.0104032
Protective effects of carbon monoxide-releasing molecule-2 on the barrier function of intestinal epithelial cells
Abstract
Objective: To investigate the protective effects and mechanisms of carbon monoxide-releasing molecule-2 (CORM-2) on barrier function of intestinal epithelial cells.
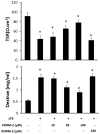
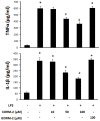
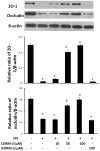
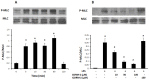
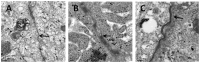
Materials and methods: After pre-incubation with CORM-2 for 1 hour, cultured intestinal epithelial IEC-6 cells were stimulated with 50 µg/ml lipopolysaccharides (LPS). Cytokines levels in culture medium were detected using ELISA kits. Trans-epithelial electrical resistance (TER) of IEC-6 cell monolayers in Transwells were measured with a Millipore electric resistance system (ERS-2; Millipore) and calculated as Ω/cm2 at different time points after LPS treatment. The permeability changes were also measured using FITC-dextran. The levels of tight junction (TJ) proteins (occludin and ZO-1) and myosin light chain (MLC) phosphorylation were detected using Western blotting with specific antibodies. The subsequent structural changes of TJ were visualized using transmission electron microscopy (TEM).
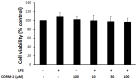
Results: CORM-2 significantly reduced LPS-induced secretion of TNF-α and IL-1β. The LPS-induced decrease of TER and increase of permeability to FITC-dextran were inhibited by CORM-2 in a concentration dependent manner (P<0.05). LPS-induced reduction of tight junction proteins and increase of MLC phosphorylation were also attenuated. In LPS-treated cells, TEM showed diminished electron-dense material and interruption of TJ and desmosomes between the apical lateral margins of adjoining cells, which were prevented by CORM-2 treatment.
Conclusions: The present study demonstrates that CORM-2, as a novel CO-releasing molecule, has ability to protect the barrier function of LPS-stimulated intestinal epithelial cells. Inhibition of inflammatory cytokines release, restoration of TJ proteins and suppression of MLC phosphorylation are among the protective effects of CORM-2.
Conflict of interest statement
Figures
Similar articles
-
Carbon Monoxide-Releasing Molecule-2 Reduces Intestinal Epithelial Tight-Junction Damage and Mortality in Septic Rats.PLoS One. 2015 Dec 31;10(12):e0145988. doi: 10.1371/journal.pone.0145988. eCollection 2015. PLoS One. 2015. PMID: 26720630 Free PMC article.
-
The carbon monoxide-releasing molecule CORM-2 inhibits the inflammatory response induced by cytokines in Caco-2 cells.Br J Pharmacol. 2007 Apr;150(8):977-86. doi: 10.1038/sj.bjp.0707184. Epub 2007 Mar 5. Br J Pharmacol. 2007. PMID: 17339836 Free PMC article.
-
[Effects of short chain fatty acid on barrier disruption of human intestinal epithelial cell induced by endotoxin/lipopolysaccharide and the related mechanism].Zhonghua Shao Shang Za Zhi. 2018 Apr 20;34(4):214-218. doi: 10.3760/cma.j.issn.1009-2587.2018.04.005. Zhonghua Shao Shang Za Zhi. 2018. PMID: 29690739 Chinese.
-
Mechanism of cytokine modulation of epithelial tight junction barrier.Front Biosci (Landmark Ed). 2009 Jan 1;14(7):2765-78. doi: 10.2741/3413. Front Biosci (Landmark Ed). 2009. PMID: 19273235 Free PMC article. Review.
-
Recent progress on anti-nociceptive effects of carbon monoxide releasing molecule-2 (CORM-2).Mol Cell Biochem. 2024 Mar;479(3):539-552. doi: 10.1007/s11010-023-04749-5. Epub 2023 Apr 27. Mol Cell Biochem. 2024. PMID: 37106243 Review.
Cited by
-
Design and Development of a Novel Peptide for Treating Intestinal Inflammation.Front Immunol. 2019 Aug 6;10:1841. doi: 10.3389/fimmu.2019.01841. eCollection 2019. Front Immunol. 2019. PMID: 31447849 Free PMC article.
-
Modulation of antigen processing by haem-oxygenase 1. Implications on inflammation and tolerance.Immunology. 2016 Sep;149(1):1-12. doi: 10.1111/imm.12605. Epub 2016 Apr 1. Immunology. 2016. PMID: 26938875 Free PMC article. Review.
-
An Overview of the Potential Therapeutic Applications of CO-Releasing Molecules.Bioinorg Chem Appl. 2018 Aug 12;2018:8547364. doi: 10.1155/2018/8547364. eCollection 2018. Bioinorg Chem Appl. 2018. PMID: 30158958 Free PMC article. Review.
-
The Protective Effect of Heme Oxygenase-1 against Intestinal Barrier Dysfunction in Cholestatic Liver Injury Is Associated with NF-κB Inhibition.Mol Med. 2017 Oct;23:215-224. doi: 10.2119/molmed.2017.00078. Epub 2017 Aug 9. Mol Med. 2017. PMID: 28805232 Free PMC article.
-
A Novel Peptide Ameliorates LPS-Induced Intestinal Inflammation and Mucosal Barrier Damage via Its Antioxidant and Antiendotoxin Effects.Int J Mol Sci. 2019 Aug 15;20(16):3974. doi: 10.3390/ijms20163974. Int J Mol Sci. 2019. PMID: 31443263 Free PMC article.
References
-
- Tenhunen R, Marver HS, Schmid R (1969) Microsomal heme oxygenase. Characterization of the enzyme. J Biol Chem 244: 6388–6394. - PubMed
-
- Maines MD (1997) The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol 37: 517–554. - PubMed
-
- Scharte M, Bone HG, Van Aken H, Meyer J (2000) Increased carbon monoxide in exhaled air of critically ill patients. Biochem Biophys Res Commun 267: 423–426. - PubMed
-
- Tsoyi K, Lee TY, Lee YS, Kim HJ, Seo HG, et al. (2009) Heme-oxygenase-1 induction and carbon monoxide-releasing molecule inhibit lipopolysaccharide (LPS)-induced high-mobility group box 1 release in vitro and improve survival of mice in LPS- and cecal ligation and puncture-induced sepsis model in vivo. Mol Pharmacol 76: 173–182. - PubMed
-
- Lancel S, Hassoun SM, Favory R, Decoster B, Motterlini R, et al. (2009) Carbon monoxide rescues mice from lethal sepsis by supporting mitochondrial energetic metabolism and activating mitochondrial biogenesis. J Pharmacol Exp Ther 329: 641–648. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

