4-Aminopyridyl-based CYP51 inhibitors as anti-Trypanosoma cruzi drug leads with improved pharmacokinetic profile and in vivo potency
- PMID: 25101801
- PMCID: PMC4148169
- DOI: 10.1021/jm500448u
4-Aminopyridyl-based CYP51 inhibitors as anti-Trypanosoma cruzi drug leads with improved pharmacokinetic profile and in vivo potency
Abstract
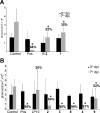
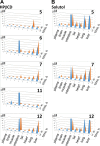
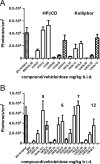
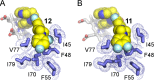
CYP51 is a P450 enzyme involved in the biosynthesis of the sterol components of eukaryotic cell membranes. CYP51 inhibitors have been developed to treat infections caused by fungi, and more recently the protozoan parasite Trypanosoma cruzi, the causative agent of Chagas disease. To specifically optimize drug candidates for T. cruzi CYP51 (TcCYP51), we explored the structure-activity relationship (SAR) of a N-indolyl-oxopyridinyl-4-aminopropanyl-based scaffold originally identified in a target-based screen. This scaffold evolved via medicinal chemistry to yield orally bioavailable leads with potent anti-T. cruzi activity in vivo. Using an animal model of infection with a transgenic T. cruzi Y luc strain expressing firefly luciferase, we prioritized the biaryl and N-arylpiperazine analogues by oral bioavailability and potency. The drug-target complexes for both scaffold variants were characterized by X-ray structure analysis. Optimization of both binding mode and pharmacokinetic properties of these compounds led to potent inhibitors against experimental T. cruzi infection.
Figures



Similar articles
-
Antitrypanosomal Activity of Sterol 14α-Demethylase (CYP51) Inhibitors VNI and VFV in the Swiss Mouse Models of Chagas Disease Induced by the Trypanosoma cruzi Y Strain.Antimicrob Agents Chemother. 2017 Mar 24;61(4):e02098-16. doi: 10.1128/AAC.02098-16. Print 2017 Apr. Antimicrob Agents Chemother. 2017. PMID: 28167559 Free PMC article.
-
Expanding the binding envelope of CYP51 inhibitors targeting Trypanosoma cruzi with 4-aminopyridyl-based sulfonamide derivatives.Chembiochem. 2014 May 26;15(8):1111-20. doi: 10.1002/cbic.201402027. Epub 2014 Apr 25. Chembiochem. 2014. PMID: 24771705 Free PMC article.
-
Binding mode and potency of N-indolyloxopyridinyl-4-aminopropanyl-based inhibitors targeting Trypanosoma cruzi CYP51.J Med Chem. 2014 Dec 11;57(23):10162-75. doi: 10.1021/jm501568b. Epub 2014 Nov 25. J Med Chem. 2014. PMID: 25393646 Free PMC article.
-
Targeting Trypanosoma cruzi sterol 14α-demethylase (CYP51).Adv Parasitol. 2011;75:65-87. doi: 10.1016/B978-0-12-385863-4.00004-6. Adv Parasitol. 2011. PMID: 21820552 Free PMC article. Review.
-
Examination of multiple Trypanosoma cruzi targets in a new drug discovery approach for Chagas disease.Bioorg Med Chem. 2022 Mar 15;58:116577. doi: 10.1016/j.bmc.2021.116577. Epub 2022 Feb 1. Bioorg Med Chem. 2022. PMID: 35189560 Review.
Cited by
-
The Potential of Secondary Metabolites from Plants as Drugs or Leads against Protozoan Neglected Diseases-Part III: In-Silico Molecular Docking Investigations.Molecules. 2016 Oct 19;21(10):1389. doi: 10.3390/molecules21101389. Molecules. 2016. PMID: 27775577 Free PMC article. Review.
-
CYP51 is an essential drug target for the treatment of primary amoebic meningoencephalitis (PAM).PLoS Negl Trop Dis. 2017 Dec 28;11(12):e0006104. doi: 10.1371/journal.pntd.0006104. eCollection 2017 Dec. PLoS Negl Trop Dis. 2017. PMID: 29284029 Free PMC article.
-
Construction and Evaluation of Molecular Models: Guide and Design of Novel SE Inhibitors.ACS Med Chem Lett. 2020 May 11;11(6):1152-1159. doi: 10.1021/acsmedchemlett.0c00017. eCollection 2020 Jun 11. ACS Med Chem Lett. 2020. PMID: 32550995 Free PMC article.
-
Utilizing Chemical Genomics to Identify Cytochrome b as a Novel Drug Target for Chagas Disease.PLoS Pathog. 2015 Jul 17;11(7):e1005058. doi: 10.1371/journal.ppat.1005058. eCollection 2015 Jul. PLoS Pathog. 2015. PMID: 26186534 Free PMC article.
-
Identification of Leucinostatins from Ophiocordyceps sp. as Antiparasitic Agents against Trypanosoma cruzi.ACS Omega. 2022 Jan 28;7(9):7675-7682. doi: 10.1021/acsomega.1c06347. eCollection 2022 Mar 8. ACS Omega. 2022. PMID: 35284725 Free PMC article.
References
-
- Chagas C. Nova trypanozomiaze humana: estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n. gen., n. sp., ajente etiolojico de nova entidade morbida do homem. Mem. Inst. Oswaldo Cruz 1909, 12159–218.
-
- Russi A. J.; Russi A.; Marin-Neto J. A. Chagas disease. Lancet 2010, 375, 1388–1402. - PubMed
-
- WHO Fact Sheet 340: Chagas Disease (American Trypanosomiasis); World Health Organization: Geneva, 2014; http://www.who.int/mediacentre/factsheets/fs340/en/ (accessed April 30, 2014).
-
- Coura J. R.; Borges-Pereira J. Chronic phase of Chagas disease: why should it be treated? A comprehensive review. Mem. Inst. Oswaldo Cruz. 2011, 1066641–645. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

